Streptomyces as a host for the secretion of heterologous proteins for the production of biopharmaceuticals
Abstract: The commercial production of therapeutic or diagnostic proteins in recombinant micro- organisms is of considerable interest. Several microbial protein production systems have been developed. So far, Escherichia coli have been the commonly employed host. However, proteins expressed in this host remain intracellular and often precipitate as inclusion bodies, which may seriously complicate downstream- processing. Faced with this problem, several genera of Gram-positive bacteria are being tested as host for the production of heterologous proteins due to their ability to efficiently secrete proteins in the culture medium. Among them is the genus Streptomyces since several of its species are known to secrete high amounts of proteins. Due to the absence of an extensive restriction-modification system, limited protease activity and the availability of suitable vector systems, Streptomyces lividans is the host of choice for the secretory production of heterologous proteins. The presented results show, that S. lividans can act as an interesting host to produce a number of proteins useful in several disease areas important in the worldwide pharmaceutical sales: i.e. oncology, immunology, cardiovascular diseases and infectious diseases.
Introduction
Streptomyces
Streptomycetes are Gram-positive soil microorganisms that produce a wide variety of secondary metabolites, many of which have potent biological activities. They produce more than half of the known biologically active microbial products, including many commercially important antibiotics, immunosuppressive compounds, animal health products, and agrochemicals [1,2,3]. They also produce various enzymes that are commercially and academically valuable as extracellular enzymes capable of degrading lignocellulosics [4,5]. This vast reservoir of diverse products makes Streptomyces one of the most important industrial microbial genera and consequently, a battery of tools for genetic manipulation of this germs is available [6]. Streptomyces coelicolor A(3)2 is genetically the best characterised Streptomyces strain. However, over the past years, Streptomyces lividans 66 or its derivatives were evaluated as potential host for heterologous protein production [7,8,9]. Secretory production of heterologous proteins by-passes the problem of inclusion body formation in the cytoplasm a problem encountered in the commonly used E. coli system. Additional advantages of S. lividans include very efficient secretion directly into the growth medium, the absence of lipopolysaccharides and simple genetic manipulation, and low-protease activity. Streptomyces lividans has been used for the heterologous secretion of several polypeptides of bacterial and eukaryotic origin. In most cases, heterologous genes are fused to signal sequences from highly expressed/secreted proteins [10].
Recombinants proteins production
The production of recombinant proteins by microbial fermentation is a main research topic in many bio-industries [11,12]. The Production of therapeutic proteins will increase 15% annually the next 5 years Examples: The worldwide sales for therapeutics IFN α (+PEGylated) will be aprox. $2700 million as Antitumor/Anti-HIV [13].
For a variety of biological and technical reasons, proteins of therapeutic or commercial interest are synthesized in either eukaryotic or prokaryotic systems. The most extensively used prokaryotic system Escherichia coli, offers several advantages including growth on inexpensive carbon sources, rapid biomass accumulation, amenability to high cell-density fermentations and simple process scale-up. But problems with inclusion body formation, rapid heterologous protein degradation and the need to purify the protein of interest free of endogenous protein often makes downstream-processing very labor- intensive and thus expensive. These drawbacks drive to search for alternative production systems. As a consequence, a variety of expression platforms has been described ranging from Gram- negative and Gram-positive prokaryotes, over several yeasts and filamentous fungi to mammalian cells [14,15,16,17].
In this study we investigated showed the feasibility of Streptomyces lividans as a host for the expression/secretion of the therapeutic proteins, human interferon alpha 2b (HuIFNα-2b). Huifna2b cDNA was fused to signal sequences of well-expressed native genes. Under appropriate fermentation conditions, significant amounts of mature biologically active HuIFNα-2b could be recovered from the spent growth media.
Materials and Methods
For expression studies, S. lividans derivatives were cultured in a suitable liquid medium (10% sucrose, 1% yeast extract, 1% glucose, 0.5% NaCl, 0.5% soya fluor, 1.7% triptone, 0.25% K2HPO4) at 27oC and 300 rpm.
HuIFNα-2b from culture supernatants of S. lividans transformed with pA∆XS, pUCIAS, pOVsiIFN or pOW15 was detected by immunoblot and ELISA according to Sánchez et al. (1998) [18].
Results and Discussion
Secretion of human interferon alpha 2b (HuIFNα–2b) by Streptomyces lividans
The interferons are a multigene family with a broad range of biological properties, including potent antiviral, antiproliferative and immunomodulatory activities.
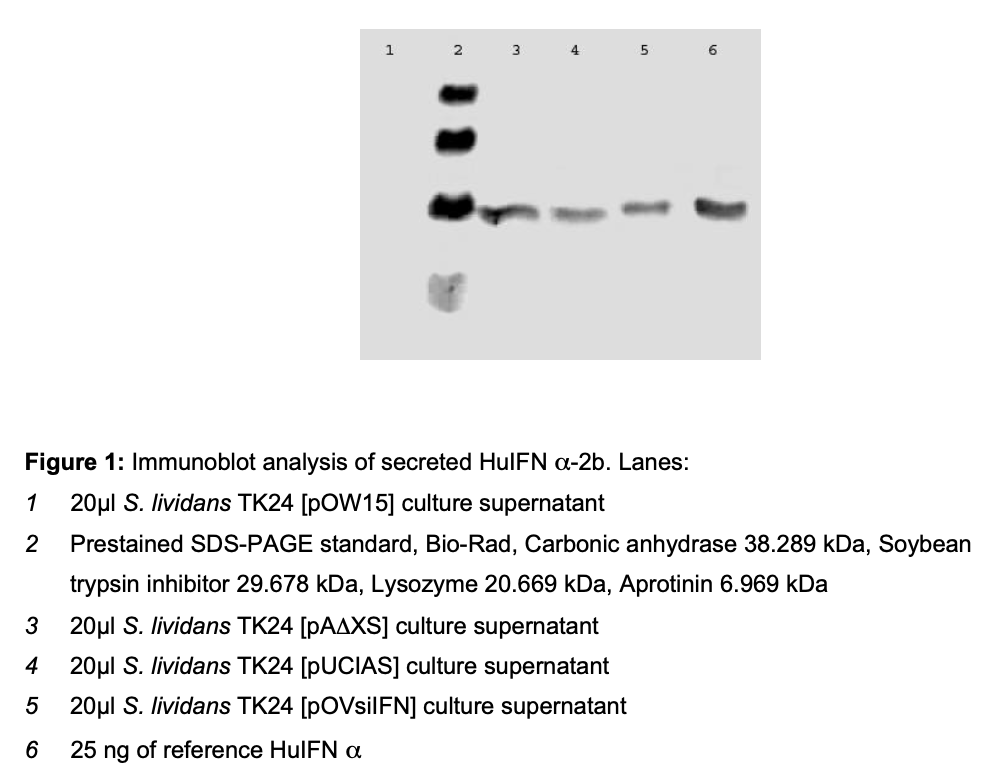
The cloning the huifna2b cDNA was achieved through recombinant DNA technology [17]. The large-scale production of this protein for therapeutic purposes using E. coli was achieved several years ago but the overexpression of HuIFNα-2b in E. coli leaded to the formation of inclusion bodies. In this study, S. lividans was used as an alternative host for the obtainment of HuIFNα-2b. Therefore the cDNA was placed under control of the regulatory sequences i.e. promoter, Shine-Dalgarno and signal sequence of Streptomyces venezuelae subtilisin inhibitor (vsi) gene or Streptomyces exfoliatus M11 Lipase A (lipA) gene. The results of the immunoblot analysis of secreted HuIFN α-2b are shown in the fig.1. The ELISA results and obtained antiviral titers of HuIFNα-2b secreted by recombinant strains of S. lividans are shown in table 1.
Plasmid pA∆XS encodes a fusion protein LipA- sp/HuIFNα–2b maintaining nine additional amino acid residues of mature LipA between the LipA signal peptide and HuIFNα–2b. [pUCIAS] encodes a fusion protein LipA-sp/HuIFNα–2b with three additional amino acid residues of mature LipA at the border of the fusion pOVsiIFN encodes a fusion protein Vsi- sp/huIFNα–2b containing two amino acid residues of mature Vsi between the Vsi signal peptide and HuIFNα–2b.
The protein HuIFNα–2b was thus expressed in S. lividans and subsequently secreted into the medium. The yield obtained for this protein and their biology activity results demonstrated the potential of S. lividans as an alternative host for E.coli aiming the production of HuIFNα-2b.

Conclusions
The presented results show, that S. lividans can act as an interesting host to produce a number of proteins useful in several disease areas important in the worldwide pharmaceutical sales: i.e. oncology, immunology, cardiovascular diseases and infectious diseases through a cost effective production systems. Although proteins of prokaryotic origin are in general more efficiently produced than eukaryotic proteins in Streptomyces lividan. Yields can be optimized using strong promoters and efficient translation signals of well- expressed native genes. Furthermore translocation signals can be modified in order to improve heterologous protein production [14]. As such for each biopharmaceutical protein a tailor-made secretion strain can be constructed.
Future developments of the fermentation processes will be investigated in order to evaluate the industrial applicability of the engineered strains and downstream processing will be optimized allowing the most efficient recovery of the secreted proteins from the culture broth.
References
[1] Goodfellow, M., and Williams, S., (1983), Ecology of Actinomycetes, Ann. Rev. Microbiol., 37, pp 189-216.
[2] Morosoli, R., Shareck, F., and Kluepfel, D., (1997), Protein secretion in streptomycetes, FEMS Microbiol. Lett., 146, pp 167–174.
[3] Connell, ND. (2001), Expression systems for use in actinomycetes and related organisms, Curr Opin Biotechnol, 12, pp 446- 449.
[4] Diaz, M., Adham, S.A., Ramon, D., Gil, J.A., Santamaria, R.I.,(2004), Streptomyces lividans and Brevibacterium lactofermentum as heterologous hosts for the production of X22 xylanase from Aspergillus nidulans, Appl. Microbiol. Biotechnol., 65, pp 401-406.
[5] Morosoli, R., Shareck, F., and Kluepfel, D., (1997), Protein secretion in strepto- mycetes. FEMS Microbiol Lett., 146, pp 167–174.
[6] Hopwood, D.A., Bibb, M.J., Chater, K.F., Kieser, T., Bruton, C.J., Kieser, H.M., Lydiate, D.J., Smith, C.P., Ward, J.M., and Schrempf, H., (1985), Genetic manipulation of Streptomyces: A laboratory manual. Norwich, C.T: John Innes Foundation.
[7] Anne, J., and Mellaert, L.V., (1993), Streptomyces lividans as host for heterologous protein production, FEMS Microbiol. Lett., 114 pp 121–128.
[8] Binnie, C., Cossar, J.D., and Stewart, D.I.H. (1997), Heterologous biopharmaceutical protein expression in Streptomyces. Trends Biotechnol., 15, pp 315- 320.
[9] Gilbert, M., Morosoli, R., Shareck, F., and Kluepfel, D., (1995), Production and secretion of proteins by streptomycetes, Crit. Rev. Biotechnol., 15 pp 13–39.
[10] Lammertyn, E., Van Mellaert, L., Schacht, S., Dillen, C, Sablon, E., Van Broekhoven, A., and J. Anné (1997) Evaluation of a novel subtilisin inhibitor gene and mutant derivatives for the expression and secretion of mouse tumor necrosis factor alpha by Streptomyces lividans, Appl. Environ. Microbiol., 63, 1808-1813.
[11] Noack, D., Geuther, R., Tonew, M., Breitling, R., and Behnke, D., (1988), Expression and secretion of interferon- alpha 1 by Streptomyces lividans: Use of staphylokinase signals and amplification of a neo gene, Gene, 68, pp 53–62.
[12] Binnie, C., Jenish, D., Cossar, D., Szabo, A., Trudeau, D., Krygsman, P., Malek, L.T,, and Stewart, D.I., (1997b), Expression and characterization of soluble human erythropoietin receptor made in Streptomyces lividans 66, Protein Expr. Purif., 11, pp 271–278.
[13] Das, R.C., (2003), Progress and prospects of protein therapeutics (application note), American Biotechnology Laboratory, October 2003, pp 8-12.
[14] Lammertyn, E., and Anne, J., (1998), Modifications of Streptomyces signal peptides and their effects on protein production and secretion, FEMS Microbiol. Lett., 160, pp 1–10.
[15] Bender, E., Koller, K.P., and Engels, J.W., (1990), Secretory synthesis of human interleukin-2 by Streptomyces lividan, Gene, 86, pp 227–232.
[16] Harth, G., Lee, B.-Y., and Horwitz, M.A. (1997), High-level heterologous expression and secretion in rapidly growing nonpathogenic mycobacteria of four major Mycobacterium tuberculosis extracellular proteins considered to be leading vaccine candidates and drug targets, Infect. Immun., 65, pp 2321–2328.
[17] Silva, A., Menendez, A., Ubieta, R., Montero, M., Torrens, I., Morales, J., Santos, A., Gonzalez, M., Jimenez, V., De la Fuente, J., Santizo, C. and Herrera, L., (1991), Expresión de Interferones Humanos en E. coli, Biotecnol. Apl., 8, 400– 405.
[18] Sánchez JC, Padrón G, Santana H, Herrera L (1998) Elimination of an HuIFN a2b readthrough species, produced inEscherichia coli, by replacing its natural translational stop signal, Journal of Biotechnology., 63, 179–186.