User research in pharma R&D: Contextual inquiry for the elicitation of user needs in a chemistry laboratory for analytical method development within a corporate continuous manufacturing organization
Abstract
Laboratories in the pharmaceutical industry see an ongoing transition towards continuous manufacturing by means of tighter integration of novel and existing technologies and, thus, the introduction of new work methodologies. However, technological studies focusing novel manufacturing methodologies usually do not address social aspects, while social sciences studies on the other hand rarely address scientific and industrial aspects of manufacturing processes and therein involved personnel. Hence, the scientific literature lacks systematic analyses of human and social factors in such continuous manufacturing environments. Therefore, the study provides a literature review of social research of scientific laboratories and lab work. Then, ethnographic field research is conducted in a laboratory for continuous manufacturing. One by one a team of six lab workers are observed and interviewed during a typical day shift (N=6). All sessions are recorded on video (4h 47mins) and transcribed to enable a qualitative content analysis. The overall work environment of a research and development chemistry laboratory of a big multinational pharma company is described including the general laboratory workflow. Finally, a list of 96 user needs as well as user role descriptions of the participating lab workers are generated. One key finding of the study is that the work culture in this lab follows a mode of constant debate trying to contain knowledge transfer in teams top-down as well as bottom-up, e.g. during experimenting with hardware setups trying not to compromise the chemical recipe following a research hypothesis. In this regard, digitization efforts like introducing electronic lab notebooks should prioritize to promote and support communication and collaboration over features and technological enhancements. Specifically, learning can be considered a shared responsibility to promote a common work process knowledge that is needed to successfully act and react in the context of continuously changing experiment setups and team compilations. Based on these results, the authors highlight the importance of holistic upfront user research to uncover underlying human and social factors as determinants for the success of socio-technical systems. All in all, with this study the authors provide a data set, which may serve as a foundation for future research and development projects in similar, industrial research working conditions, following a human-centered design approach.
1 Introduction
Social research addressing the pharmaceuti- cal industry or the life sciences is usually focused on high-level critical debates about political regulation, consumption and consumerism, customer expectations and broader innovations (Williams et al., 2008), while social studies that address scientific and industrial aspects of manufacturing processes and the therein involved people are rare. This assertion is true for processes in corporate research and development (R&D) in industrial research (Darrouzet et al., 2009; Jordan and Lambert, 2009) and it is especially true for R&D in the pharmaceutical industry and the life sciences industry. Typically, such R&D organizations are focused on the creation of intellectual property (IP) as the output of all activities. Some pharmaceutical researchers argue that this strict focus is the root of the “culture of secrecy” and a cause for the massive duplication of effort. Further, these researchers argue for the adoption of open science approaches allowing organizations to experiment with new forms of collaboration (Bountra et al., 2017). This is also what makes this study unique, because social field research in many cases still lacks recognition of management and business stakeholders. As a result, social scientists, ethnographers and user researchers in industry often find their efforts devalued, neglected, or not realized by stakeholders (Amirebrahimi, 2015).
In this spirit of innovation by means of openness and transparency this study aims to encourage a more deliberate discussion about laboratory processes. Specifically, these strict R&D processes center around highly regulated laboratory workplaces, which are common place in any pharmaceutical or life sciences organization (Osakwe, 2016).
However, there is a lack of research that offers insights into how these corporate labs are run, how people actually work there, what these people require to work efficiently, and what they might need in the future to enhance their current workflows.
Now, this study aims to shed light on the field of corporate laboratory work by investigating the work performed in a research laboratory for continuous manufacturing of a big, multinational pharma company.
1.1 Background: Working in pharma R&D
The pharmaceutical industry includes the manufacture, extraction, processing, purification, and packaging of chemical materials to be used as medications for humans or animals (World Bank Group, 1998; Konstantinos et al., 2011). Following Konstantinos and colleagues (2011), pharmaceutical manufacturing can be separated into two main phases: (a) the production of the active ingredient or drug (primary processing, or manufacture) and (b) the secondary processing, the conversion of the active drugs into products suitable for administration.
According to the European Federation of Pharmaceutical Industries (EFPIA), turning a newly synthesized active substance into a marketable medicinal product takes an average of 12-13 years. However, only three in ten of these products will produce revenues that more than cover their research and development costs (EFIPIA 2010; Konstantinos et al., 2011). In addition, the pharmaceutical manufacturing industry produces therapeutic substance (human and veterinary medicines, drugs, and related products) in an increasingly concentrated set of mostly transnational company and sub-contracting facilities. The sector has five broad areas of activity: (a) research and development, (b) manufacturing, (c) sales and marketing, (d) distribution, and (e) administration (Konstantinos et al., 2011).
For the manufacturing of pharmaceutical products many facilities have multi product capability and the equipment may in some cases be the same as are operating personnel. Thus, in the same workplace different raw materials are used, different processes are executed, and different waste streams are generated (Gad, 2008; Konstantinos et al., 2011). These facilities are considered highly maintained environment wherein equipment must be cleaned, to avoid cross-contamination. This involves water, steam, detergents, as well as organic solvents.
Today, many steps are automated in these processes, with examples of employee tasks including: (a) weighing and dispensing solids and liquids (using pumps or pouring), (b) charging and discharging solids and liquids from containers and process equipment, (c) manual materials handling, (d) equipment maintenance and repair, and (e) watching controls and processes (Konstantinos et al., 2011).
Furthermore, as Konstantinos and colleagues (2011) summarize, the employees working in these environments of a manufacturing facility may be exposed to all kinds of influences like noise, heat, and humidity. Also, surfaces can be hot and slippery, while some surfaces and floors may be covered with dust from the process. Moreover, employees may be exposed to hazards like moving machinery parts and pressurized pipes and vessels, and some work is done in confined spaces or with high-energy sources. In extreme circumstances involving large quantities of highly charged powder particles explosive atmospheres can exist, for example, solvents can burn or explode, especially in organic synthesis. To cover all these work-related risks, general manufacturing practice and other quality control rules set by regulatory agencies, customers, and pharmaceutical organizations cover a number of these processes and the equipment used. Health and safety laws as well as good manufacturing practices (GMP) guidelines apply to all of them (Konstantinos et al., 2011).
Overall, the pharmaceutical industry relies on highly-regulated work processes on the one hand, while it relies on constant discovery of new recipes for drugs in R&D that may have great impact on the health of consumers on the other hand. The main goal of the laboratory is to gain a fundamental understanding of chemi- cal synthesis through workflows of mechanistic and statistical analysis, ultimately leading to increased knowledge of not just one process, but of common schemes.
1.2 Current developments: From batch to continuous manufacturing
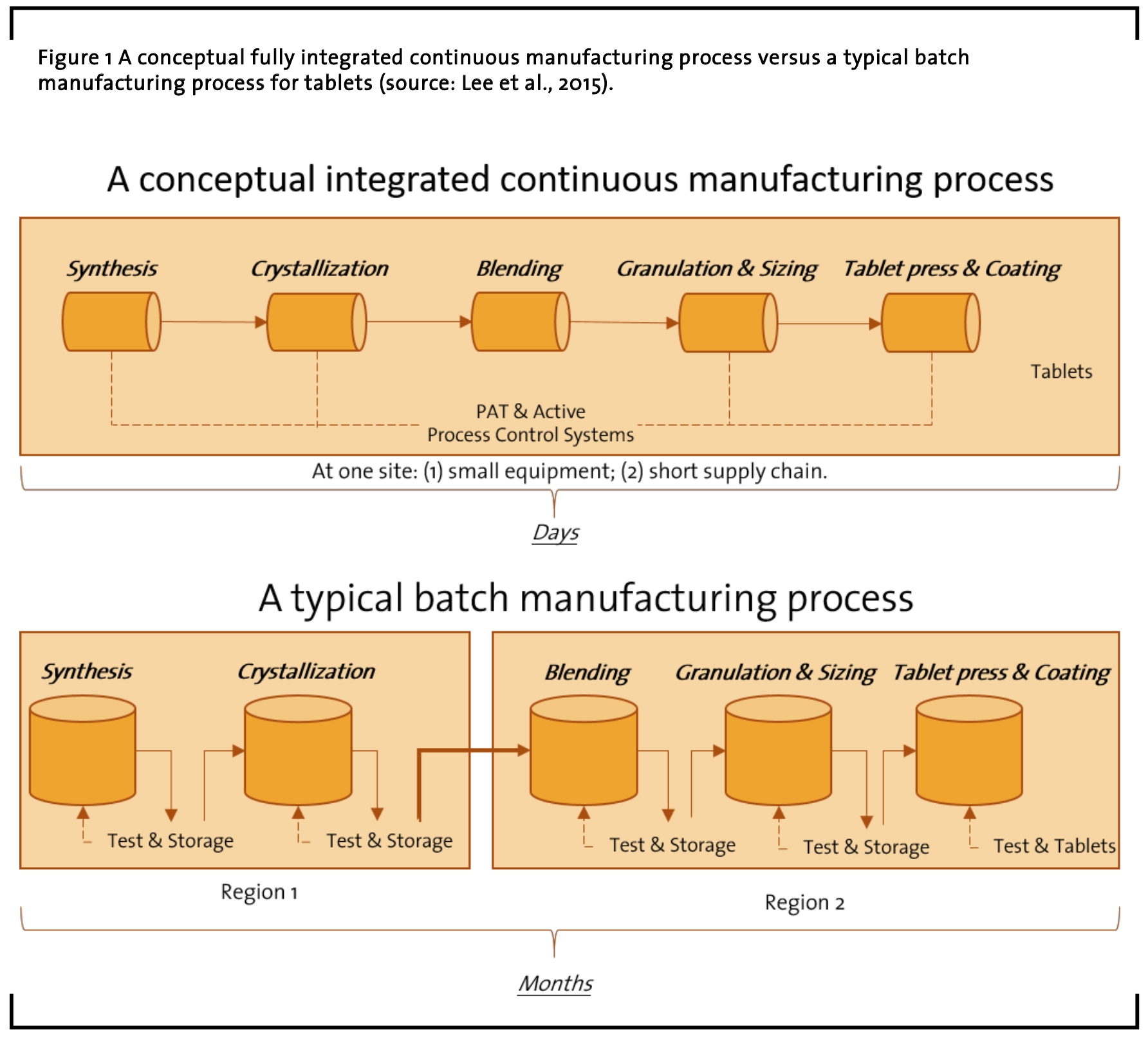
For several decades, pharmaceuticals have been produced using a method known as ‘batch manufacturing’, a multi-step, lengthy process that involves the use of ungainly, large-scale equipment (see Figure 1). Recently, advances in manufacturing technology have encouraged the pharmaceutical industry to move from this traditional way of manufacturing to a faster, more efficient process known as ‘continuous manufacturing’ (FDA, 2004; Lee et al., 2015). Some authors even argue that the lack of agility, flexibility, and robustness in the pharmaceutical manufacturing sector poses a potential public health threat as failures within manufacturing facilities that result in poor product quality can lead to drug shortages (Throckmorton, 2014; Myerson et al., 2015; Lee et al., 2015). In this regard, the Food and Drug Administration (FDA) considers continuous manufacturing the innovation that has great potential to improve the aforementioned level of agility, flexibility, and robustness in the manufacturing of pharmaceuticals (Lee et al, 2015).
In continuous manufacturing pharmaceuticals are moved nonstop within the same facility, which eliminates hold times between steps. Substances are fed through an assembly line of fully integrated components, which saves time, can reduce the likelihood for human error, and lab managers can respond more easily to market changes.
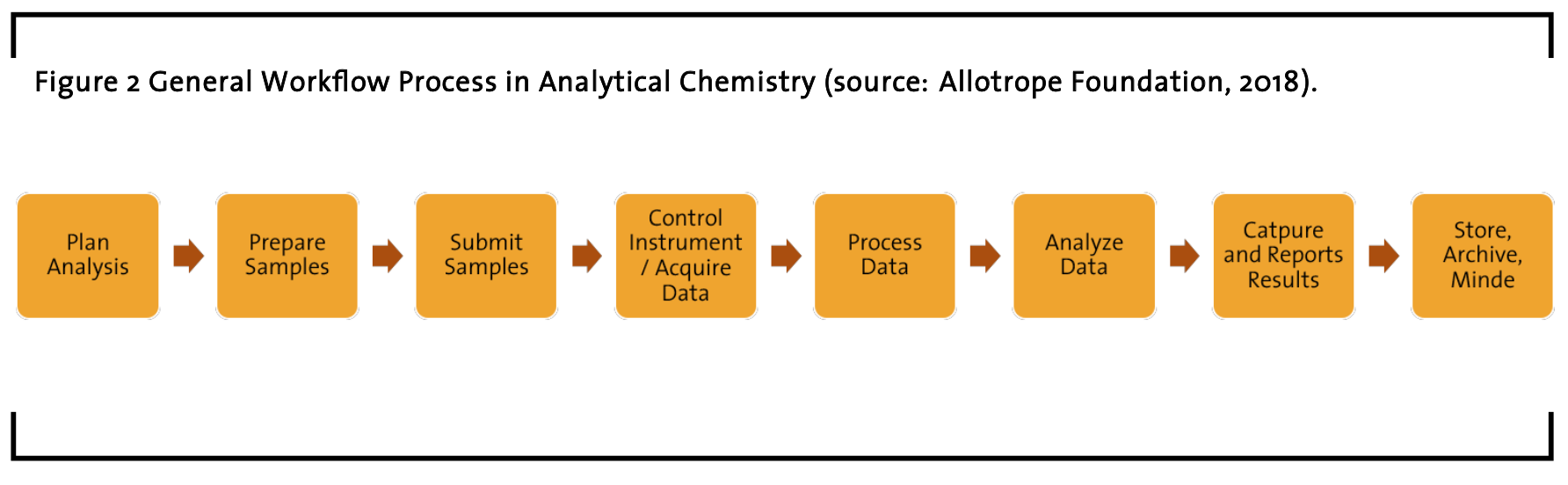
However, continuous manufacturing requi- res a different experimental approach. The impact of the equipment used within drug development has a large impact of the industrialization approach, which requires far greater understanding of the impacts of the equipment on the synthesis of substances. Specifically, certain equipment does not scale, especially those used in early screening, such as microfluidics. Therefore, a complete understanding of the characteristics of the equipment is needed to ensure that the processes are well understood to realize commercial requirements. Done right, the coupling of mechanistic process understanding along with equipment models allows the development process to move rapidly from different scales of operation, without the need for extended development operations. To ensure that the required process data is captured to support the mechanistic model generation, standard platforms are needed to provide consistent data. These platforms have a high level of automated control, data capture and data processing. In this regard, debates about standards of workflows, process models and data handling are still ongoing, e.g. in international consortiums like the Allotrope Foundation (see Figure 2). While continuous manufacturing is a general methodology, the investigation of a laboratory for so-called ‘analytical method development’ following this methodology builds the center of this study. Therefore, this study aims to investigate the people and their work in pharmaceutical research and development laboratories for continuous manufacturing.
1.3 Literature review
In order to research people working in pharmaceutical research and development laboratories for continuous manufacturing a literature review is conducted. Its goal is to review current literature that studies cultural, social and psychological factors in laboratory work. The review applies the following two major literature categories: Academic Social Sciences literature originating in anthropology, sociology, and general psychology; and Applied Sciences Literature originating in (a) education and instructional psychology, (b) human factors, and (c) user experience design.
Academic Social Sciences Literature about Lab Work
The academic social science studies of scientific laboratories go back to the 1970s and is called ‘Science and Technology Studies’ (STS). Typically, the main aim of laboratory studies in STS addresses broader concerns of epistemology of science and technology demonstrating the local accomplishment and social construction of scientific knowledge (Harrington, 2013; Sormani, 2014; Stephens and Lewis, 2017; Friberg, 2017). The research perspective in these studies is committed to the interactionist tradition of ethnographic work that focuses on the interactive practice and detailed observation of how scientific work is accomplished through social interaction (Atkinson et al., 2008; Atkinson, 2015; Stephens and Lewis, 2017). Other approaches to laboratory studies are rooted in sociology which treated scientific practice as a strange and alien culture (Latour and Woolgar, 1986). This is in contrast to other research focusing on understanding the scientific work from the individual perspective of the scientists (Knorr, 1977). But all authors utilized the detailed ethnographic observation of day-to-day work to document how normal scientific knowledge is accomplished (Knorr, 1977; Latour and Woolgar, 1986; Lynch, 1985; Traweek, 1988). Somehow connected to this research are organizational studies investigating the every-day life, skills, knowledge, identities, and attitudes of technicians as a general working type (Barley, 1996; Barley et al., 2016).
Another body of laboratory ethnography specifically investigates scientific laboratories as spatial arrangements and work environments for the social construction of knowledge. This research also borrows from social research of architecture design (Gieryn, 2002). Here, the laboratories are treated as special work spaces focusing on rhythms of day-to-day work, movements, materials, transitions, boundaries, and barriers as labs come in different shapes and sizes. Some are large and spacious, others small and confined, some are busy and heavily populated, others quiet and conspicuous by the absence of workers (Stephens and Lewis, 2017). Many labs are gated communities, others are linked closely to hospitals and clinics. Some are distant from highly populated regions e.g., the UK Stem Cell Bank (Stephens et al., 2008), and some are transient spaces that pop-up as “portable packages” (Lewis et al., 2014; Ste- phens and Lewis, 2017). In this regard, some authors argue for a radical redesign of scientific space speculating that new life science buildings following these redesigns are designed not only to intentionally produce intense social action between scientists but are also built to reflect the state and status of bioscientific innovation (Thrift, 2006). Other researchers argue for shifting focus of STS studies towards the private sector (Penders et al., 2009).
Applied Sciences Literature
The body of research coming from applied sciences, industries, and corporations aims to investigate and optimize the work experience for employees (e.g., training and ergonomics), workflows, technical processes, management, and businesses.
Education and Instructional Psychology
One branch of research coming from the disciplines of education and instructional psychology discusses current challenges like lifelong learning on the job or mental stress in knowledge work (Boreham and Morgan, 2004; Fischer, 2005; Grundgeiger et al., 2017; Bahl and Dietzen, 2019). Here, some research is focused on learning at work in general (Maclean et al., 2009; Fischer et al., 2004) as well as learning and teaching in specific domains of work (Boreham et al., 2002; Fischer, 2005). Some studies include learning in laboratory work environments with respect to the generation and appropriation of ‘work process knowledge’ of chemical laboratory assistants in particular (Talanquer, 2006; Fischer and Röben, 2002a; Fischer and Röben, 1997; Storz et al., 1997; Kruse, 1986). Following Boreham (2002) the concept of ‘work process knowledge’ goes back to Kruse (1986) who originally defined the term as ‘labour process knowledge’, meaning:
- an expanded understanding of work roles in parts of the organization other than the employee’s own;
- an awareness of the interdependency of the activities in different departments, including characteristics of the system as a whole, such as the flow of work through the organization, both upstream and downstream of the worker’s own station; and
- participation in a workplace culture which provides a service to colleagues in support of a high quality of service to the actual customer.
The concept of work process knowledge was developed to define the knowledge that workers, whether hotel employees, machinists or laboratory assistants, need in order to cope with more organic and knowledge-creating working environments. Developing work process knowledge helped them to adjust to more flexible processes (Boreham and Fischer, 2009; Fischer and Boreham, 2004).
However, only a few studies investigate learning processes inside scientific laboratories of companies (Torz and Eichhorn, 2001; Röben et al., 1998). Mariani (2002) as an exception conducts an in-depth investigation of the R&D practices of a major Italian chemical company that has been in existence for 40 years and employs about 1000 people. The study is focused on investigating the claimed competitive advantage of this company by looking into team work and the respective work process knowledge of individual team members, while including organizational factors of short-term contracts for learning and group dynamics. In this study the site management of the company is convinced that innovation and especially the invention of new products are a function of the number of tests and experiments that are run. Research processes, in contrast to production processes, have very uncertain outcomes and it can be very difficult to make a rational selection of trials. Hereby, one comment stands out pointing towards the special circumstances and social dynamic of laboratory teamwork:
“When the R&D project is planned, managers of the different customer areas and researchers are asked their ‘wish list’ of activities. They always say that they need a research centre that is three times bigger. But when the R&D starts, we find that the eighty percent of the new ideas originate from everyday activities on the basis of local insights. (Excerpt from an interview with the person responsible for Planning and Control)” (Mariani, 2002)
The employees of the chemical company argue for flexibility of workflows within a strict ruleset of controls for scientific discovery and safety regulations. Therefore, Mariani (2002) concludes that successful R&D depends critically on two preconditions: The capacity to perform a large number of experiments and the capacity to rapidly modify or adjust production programs so that, whenever a line of research produces promising results, processes in the plant focus on producing the chemical in question. Interestingly, the call for flexibility and increased rate of experimentation over regulations to foster scientific discovery is also demonstrated by changes in attitudes towards confidentiality. During the evolution of this chemical company’s laboratory work culture the employee’s focus shifted from covering all site installations with opaque steel shielding to get rid of everything that slows down the whole process, including expenditure for hiding their work. Employees summarized this shift in philosophy by stating that if they are first, it would not matter who might copy them. Regarding the organization of the company’s lab work both the pilot plants and the laboratories started to operate on a 24-hour cycle in order to maximize the rate of experimentation. Also, on the basis of a very small increase in personnel, activity in the pilot plants was increased from one experiment per month at the beginning of the 1980s to the present rate of two per week. And by introducing a continuous work cycle, the output of the laboratories was increased from two or three tests per day to 20 to 25 per day.
These intense boundaries for teamwork seem to be especially effective in the pilot plants of the company. Work in these pilot plants consists mainly of running investigations to discover new products and develop new production technologies. As Mariani (2002) observes, this work is managed by two types of teams, one performing on a technical level and one on an operational level. The technical teams are responsible for setting up and modifying the program in each pilot plant. They consist of three people with different responsibilities:
- a plant manager who takes responsibility for the overall functioning of the plant (this role was created in the early 1970s),
- a process engineer, responsible for desig- ning the test which is to be run (this role was created in the mid 1980’s),
- a technologist responsible for the technolo- gy needed to perform the test (this role was created in 1990). Its rationale is that, to adjust activities as a function of progressively emerging findings, it is critical that the technological configuration of the scaled down chemical installations can be altered very rapidly.
In order to investigate the lab work in teams of such chemical companies it is important to be aware of technical constraints in contrast to the adaptability of chemical formulas. For example, while the time required by the investigation itself might be fixed, the time needed for installation set up could be shortened as a function of team efficiency (Mariani, 2002). In this regard, team members of this chemical company’s operational team rotate between roles to secure efficient teamwork even if one team member drops out. This way the lab team is flexible enough to react on changing circumstances. In addition, this kind of rotation exists for technical employees, too. Before a new employee, such as an area technologist, becomes fully operational, he or she is asked to work for a period in the operational teams as an ordinary team member. As Mariani (2002) concludes, this eliminates the familiar problem of engineers and researchers who are very knowledgeable theoretically, but whose knowledge of plant structure and organization is almost nil. Within both teams, therefore, everybody comes to know everybody else’s job and to compensate each other’s lack of experience. In this way, the different phases of the work process become well known to all members of the team, making it possible to develop a common language that allows a tight coupling of activities. Beyond this idea of sharing work process knowledge to ensure efficiency, having experts to teach their work to novices can be regarded as very positive alone (Mariani, 2002). It represents a step back to the tradition of apprenticeship that has been undermined by industrial change or as one experienced lab worker commented during the study (Mariani, 2002):
“It also . . . increases our professionalism. For example, if we did not have to teach some of our plant schemes to newcomers, we would not look at those schemes for four-to-five months. In this way, we have got to continuously refresh our knowledge in order to be able to transmit it. (Excerpt from an interview with a skilled worker)”
However, the informative corporate study of Mariani (2002) does not offer concrete descriptions of the actual lab work (workflows) nor does it offer insights into the specific needs of lab workers (user needs) to work in a team and to perform respective everyday tasks. So far it can be noted that laboratory workers possess a specific kind of work process knowledge that is quite different from academic knowledge of chemistry and that dynamic team work in some laboratories has high demands of situational adaptability. Nevertheless, their work is similar to work in any scientific lab: Variations are introduced in existing substances by a (synthetic) chemist, which are subjected to standardized tests to determine their chemical properties.
Now, complementary to Mariani (2002), Fischer and Röben (2002a) investigate the actual work of laboratory assistants inside analytic laboratories. Analytic laboratories are involved in both the development of new drug substances eventually becoming medicines and the translation of the chemical reaction from the laboratory standard to the production standard. For example, such labs are responsible for determining the structure, purity, and content of substances produced by means of synthetic chemists. They develop the first procedure for ascertaining the identity, purity, and content of screening substances. Also, these laboratories offer advice with regard to possible ways of synthesizing substances. In addition, standard operating procedures for determining the identity, purity, and contents of the byproducts of these syntheses are elaborated and stability tests, in particular, are carried out. Regarding the work environment and workflows, the authors investigate the organizational structure of labs. They postulate that the fundamental principle of the organization of a chemical laboratory is the established division of labor between a chemist and a laboratory assistant, wherein, traditionally the latter is considered the former’s helper (Fischer and Röben, 2001).
Specifically, in the companies the authors surveyed, the employees of the laboratory work in teams mostly made up of one chemist and several laboratory assistants. These teams work on one or more analytical procedures while the number of people working in a team depends on the amount of work needed for the analytical methods used, and also on the complexity of these methods (Fischer and Röben, 2002a; Röben, 2002b). Furthermore, the authors describe the different responsibilities of scientists (e.g. chemists or pharmacists) and assistants. Work of scientists is based on their scientific knowledge about the structures, reactions and properties of chemical substances (Schmauderer, 1973). Sometimes scientists are acting as heads of a laboratory and manage teams which usually consist of two to five laboratory employees. More often scientists are taking charge of research projects, which includes that they discuss and collaboratively decide what is to be measured, in what form and with which methods (Fischer and Röben, 2002a; 2002b). This academic laboratory staff is occupied with planning and carrying out analysis as well as evaluating and controlling validity of results.
The work of laboratory assistants on the other hand is focused on determining the quality and the quantity of substances (Ciommer, 1996; Fischer and Röben, 2002a). While a chemical analysis in its entirety goes through the stages of taking samples, preparing samples, analysis and evaluation, the job of laboratory assistants consists mainly of the preparation of samples and then the process of taking measurements (Mohler, 1970). Generally speaking, they work in the context of the work of scientists, that is, cleaning components and the lab environment, maintenance, calibration, quality control, buying equipment, taking samples, or providing and documenting measurements at the end of an analysis. Of course, this assisting work is critical to the quality of the measurement, which is – in most cases – done automatically by computer-controlled instruments (Fischer and Röben, 2002a). But this does not relieve the lab assistant’s work, because the representativeness of samples must not be compromised by any treatment carried out before the computer-controlled measurement. Thus, meeting these conditions requires working closely to the instructions and regulations, which represent the context of the analysis. The authors stress the fact that, for example, scientific procedures given for an analysis might specify that a sample should be crushed with a pestle and mortar, dissolved in a solution and then filtered. But these procedures do not say anything about the method of filtering, which is not a trivial issue. Therefore, it is argued again that laboratory assistants need a kind of work process knowledge to successfully interpret and follow laboratory instructions (Fischer and Röben, 2002a). Here, the authors rely on two psychological constructs: Context awareness and context comprehension. The authors describe work process knowledge as a construct that connects the requirements of a task with the company-specific conditions under which a task is to be carried out (Boreham et al., 2002). Such knowledge is acquired from the experience gained by working in a specific work environment. This is important, because it is often the case that for example an instruction refers to a five minutes treatment of a sample, while practical experience tells that this treatment might require eight minutes with a particular device. Another example are process drawings or specifications, which are usually made by someone other than the assistant worker who uses it. Here, the lab assistant needs to learn and understand the specifications before he or she uses it in the work process. This insight then follows the argument that if a laboratory assistant lacks the relevant scientific understanding, it is not possible to develop adequate work process knowledge (Fischer and Röben, 2002a). Successful lab work relies on subject matter expertise, practical expertise as much as it relies on the actual work community and the division of labor. Therefore, like Mariani (2002) the authors conclude that it seems to be very important that lab assistants are able to make mistakes within the rule set of strict organizational regulations, in order to educate themselves in a pragmatic learning-by-doing manner within a scientific laboratory environment to progressively combine their practical knowledge with the feedback and scientific knowledge of their colleagues (Fischer and Röben, 2002a).
Human Factors Literature
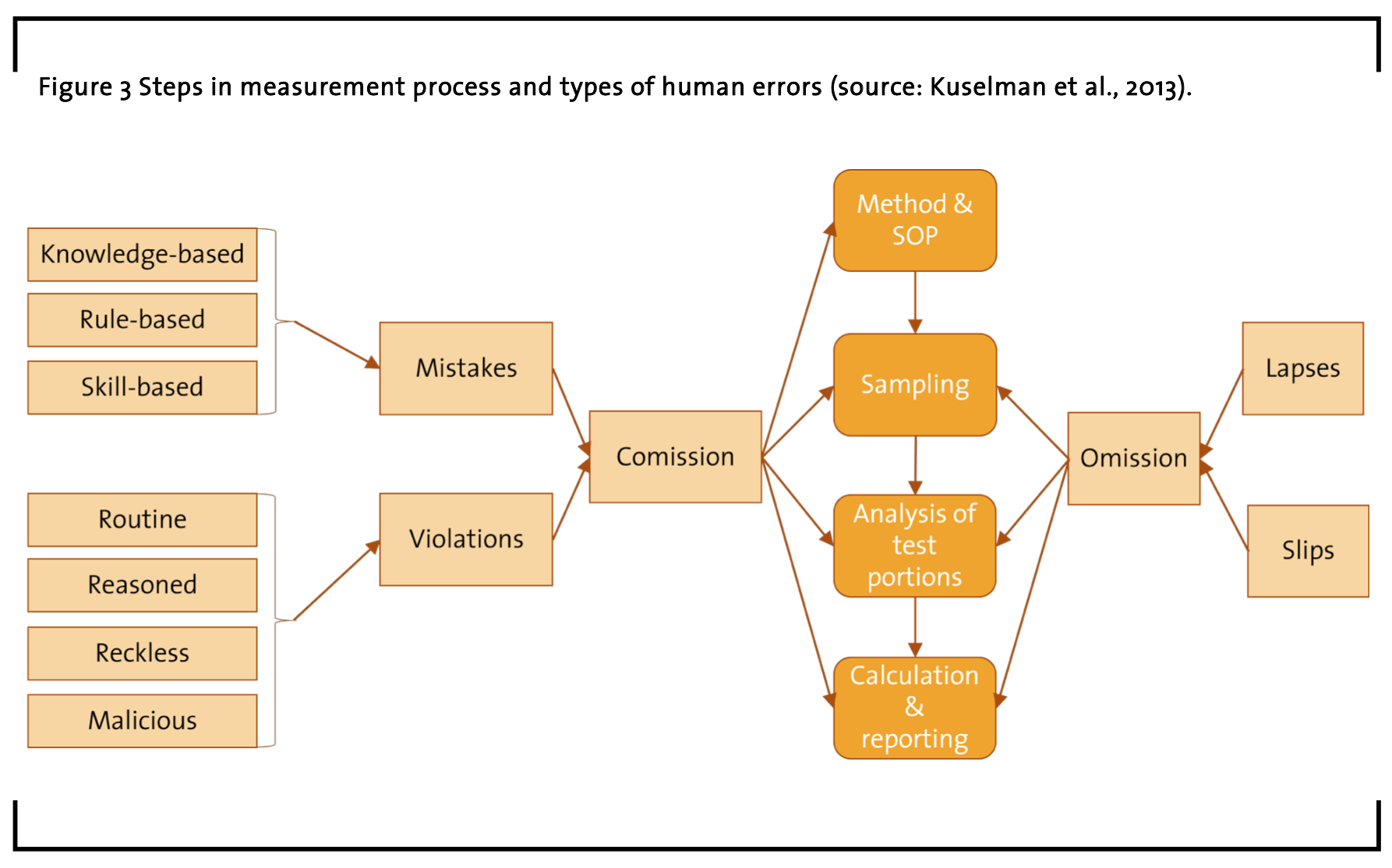
In contrast to this body of applied research about scientific laboratories focusing on education, training and instructional psychology, there is another prominent field of applied research focusing on optimizing work processes. The Human Factors and Ergonomics (HFE) studies conducted on scientific research laboratories as a work domain are minimal (Jones, 2005; Jones and Nemeth, 2004). This is probably because historically HFE as a discipline has emphasized naturalistic studies of real-life “in the wild”, in order to contextualize laboratory-based experimental results (Kant and Burns, 2016). Besides the interdisciplinary nature of HFE that as a discipline proposes a holistic view regarding product strategy, field research, requirements engineering, and usability engineering (Privitera, 2019), another general theme in HFE research is the strong focus on human error (see Figure 3; e.g. false identification of a component) in contrast to equipment error (e.g. insufficient sample or a broken container), and safety issues (e.g. individual attitude damage or hygiene regulations). This is also true for HFE studies about laboratory work environments (Ala and Bagot, 1994; Bonini et al., 2002; Hockey, 2005; Ross, 2008; Haile and Hussen, 2012; Kuselman et al., 2013; Perry, 2018). Here, HFE researchers try to transfer their experience and knowledge from work on medical devices, military systems, and aviation safety systems (e.g. Endsley and Robertson, 1996) to the lab work (Konstantinos et al., 2011).
Nevertheless, it is argued that HFE research could provide sustainable mechanisms to support innovation at a ‘‘grassroots-level’’; i.e., from ‘‘bottom-up” (Kant and Burns, 2016). Another limiting aspect of these initial HFE studies is the fact that they typically aim to study laboratory processes exclusively addressing scientific laboratory work within government research facilities or universities. Access to corporate research facilities seems to be an issue. However, in this regard the study of Konstantinos et al. (2011) can be highlighted as a resourceful source of insights. The study is focused on Human Factors, that is, any factor that affects human performance and increases the probability of errors in the workplace. Theoretically, the authors base their work on a model created to represent the human factors in aviation maintenance. This model was named Safety Training for the Aircraft Maintenance Industry. The study of Konstantinos and colleagues (2011) contains a literature review of legislation surrounding the manufacturing process of pharmaceuticals (Good Manufacturing Practice, GMP; Occupational Safety and Health, OSH), so as to reveal if human factors were incorporated in the various laws, directives or guidelines produced by the legislative bodies. Secondly, the authors conducted research at a large commercial biopharmaceutical manufacturing facility and a small pharmaceutical manufacturer.
Overall, the results of their literature review show that the compliance with the legislation regarding the GMP regulations are the main requirement for pharmaceutical production. This does not directly emphasize the human factors aspect. On the other hand, the authors conclude that the researched OSH legislation gives more attention to human factors, although indirectly, and could be used as guidance towards effective organizational planning that incorporates more of these aspects.
Here, the authors conclude that if an organization uses both GMP and OSH legislation to their full effect, it will provide the foundations for a safe, dynamic working environment that has as the production of safe pharmaceutical products of high quality as main priority, but also respects the individual needs and abilities of the worker. Thus, the likelihood for errors produced from human factors will be reduced and their effect on manufacturing minimized. Furthermore, the authors mention the future trends in pharmaceutical manufacturing and in industry as a whole, that reinforce the need of a stronger utilization of human factors aspects, as many issues of concern can be avoided or their effect minimized (Konstantinos et al., 2011).
In addition to the literature review, the authors conducted observations in the field (what they call “walk-arounds”) to identify cognitive demands, physical demands, verbal communication requirements, non-verbal communication requirements (e.g. paperwork), interaction with automation and equipment, potential human errors, potential machine errors, potential human-machine interaction error, and general workload issues. Also, they conducted informal one hour interviews to investigate responsibilities, priorities, rules, staff, working hours, sources of pressures and constraints, and work-arounds. In addition, the authors conducted a survey to gather perceptions, opinions and further comments regarding factors that influence human performance and endanger worker safety, reliability, and pose a risk to the product. The survey was made available at lar- ge scale to employees at all levels and all departments.
The final results contain an overview of “skills/competencies required” in the daily work of the study participants, which represent “factors that can lead to errors/events in BPM” from the authors Human Factors perspective. For example:
(a) Task-related skills include “Preserving at- tention to detail”, “set up and clean processes”, “responding to time/operational/production pressures”;
(b) Individual-related skills include “Physical, cognitive capabilities and limitations on job performance (dealing with fatigue, interruptions, distractions, complacency, etc.)”, “identifying the preconditions/precursors for errors”, “Patience: resistance to rushing; maintaining vigilance”, “Professionalism: sense of ownership in task/process/product-outcome”;
(c) Team-related skills include “Teamwork (benefits, challenges)”, “Danger of diffusion of responsibility”, “Communication (effective, frequent, different types of, as a function of level, of time of day, limitations of)”, “Hand-over/ shift changes: ensuring situation awareness/ common operational picture quality support needed on shifts, transfer of responsibility, planning activities near hand-over”;
(d) Organization-related skills including “Management of unscheduled-tasks”, “Monitoring (without losing focus)”, “Cross training (advantages, disadvantages)”, “Introduction of new technologies/equipment”, or “Time-Management” for example (Konstantinos et al., 2011).
In addition, the survey results contain several issues regarding risks for human error. Specifically, as far as task factors are concerned, the primary human factors-related issues that employees generally agree are potentially error-inducing and threaten safety: (a) Time Pressure, (b) Distractions, and (c) Interruptions. Furthermore, “Unclear procedures” was reported as an issue that appeared to specifically concern employees, perhaps due to a large range of activities which require detailed procedures. Konstantinos and colleagues (2011) see a trend for employees to believe that the human factors issues ranking high on the list of concerns are also those for which employees are not well-equipped or trained to effectively address them. Regarding individual factors, the respectively same issues seemed to concern employees: “Stress,” “Fatigue,” and “Personal problems”. The authors mention that some employees reported that they had some type of training on how to handle fatigue, while all respondents appear to believe that “Stress” is an issue for which they require more assistance in dealing with. In addition, survey participants from the larger facility placed emphasis on the issues of “Lack of motivation” and “Personal problems” (Konstantinos et al., 2011). On the other hand, both team and organizational factors appeared to generate more consensus regarding their potential to induce errors. Half of the listed team and organizational issues generated agreement that they are potentially error-inducing, compared to only a third of the listed task-related issues and individual issues. Within team factors, specifically, the issue that appeared to concern all participants the most were unclear roles and responsibilities. Another issue the study showed was a general unwillingness to ask for help from colleagues. All in all, the top-issue from all target groups and facilities indicated not having been trained on how to deal with this lack of communication among team members. Lastly, within Organizational factors, respondents from different facilities responded differently about issues that may lead to errors. At the smaller facility respondents selected: (a) Lack of effective response from Supervisors and Management regarding reported safety issues, while respondents from the larger facility selected, (b) Insufficient workforce, (c) Inadequate tools and equipment, (d) Poor documentation as issues which may lead to error. In terms of knowing how to handle issues, the interesting finding has to do with the larger facility response, which highlights the need for training regarding a clear leadership structure (Konstantinos et al., 2011). All in all, the study from Konstantinos and colleagues (2011) follows the tradition of HFE research focusing on the specification of human errors in specific situations, while concrete workflows and observations are not described in detail.
User-Experience Design Literature
Following, the literature coming from User Experience Research is discussed (UXR; formerly called User Research or Usability Engineering). UXR is another field of disciplines that is in relationship with Human Factors and Ergonomics. But, while HFE is rooted in occupational and engineering psychology (Badke-Schaub et al., 2008), the quite younger discipline of User Experience Research goes back to the human-centered design methodologies co-developed by psychological researchers and design practitioners within the fields of Human-Computer Interaction (Stuart et al., 1983; Norman, 1988; Preece et al., 2007; Grudin, 2017) and Product Design (Holtzblatt and Beyer, 2017) hereby the authors include the fields of industrial design, media design, user interface design, graphics design, and visual design). Although there are a lot of studies of industrial environments and systems (Lee et al., 2017; Jakl et al., 2018; Aro- maa et al., 2018; Karim and Tretten, 2014; Terzic et al., 2009) and some exclusively focus on user research to explore user needs (Palviainen and Leskinen, 2006; Sørensen et al., 2008), techno- logy adoption (Singh, 2019) or evaluation of user acceptance (Gavish et al., 2015), there seems to be not one study regarding the user needs within scientific laboratory work environments.
Conclusion and Research Question
Social research addressing the pharmaceutical industry or the life sciences is usually focused on high-level critical debates about political regulation, consumption and consumerism, customer expectations and broader innovations (Williams et al., 2008), while social studies that address scientific and industrial aspects of manufacturing processes and the therein involved people are rare. This assertion is true for processes in corporate R&D in industrial manufacturing (Darrouzet et al., 2009; Jor- dan and Lambert, 2009) and it is especially true for R&D in the pharmaceutical industry and the life sciences industry.
Currently, social research of laboratory work lacks systematization from an interdisciplinary point of view. Studies aiming for an academic audience of social scientists have a diverse and broad set of topics and are very specific in their theoretical perspective, e.g. investigating the spatial arrangements of laboratories. The Applied Sciences literature on the contrary seems to have a limited set of topics that share general themes of process optimization and efficiency, e.g. focusing on identifying the potential for human error in general laboratory workflows. Another aspect is the clear lack of studies on lab work coming from the human-centered design community, although comparable studies investigating work inside of other industries seem promising, especially in view of hedonic qualities of actual working experience (see: User Experience Design). However, many studies of human-centered design are performed privately as part of contract work and may not be published respectively.
In sum, the current literature either explores fundamental themes like the social construction of scientific knowledge or identifies and discusses specific human factors in an explanatory way trying to understand cause and effect of errors and respective risks in manufacturing. While individual studies are exploring processes of learning and knowledge sharing of teams working in laboratories, they do not explicitly describe the actual workflows, tasks, responsibilities, expectations and respective needs of people.
In addition, there seems to be a general selection bias in the actual access to laboratory facilities that limits the scope of investigation, e.g. while the study of work in public labs of universities seems to be convenient, only a few publications address the work in corporate laboratories.
Therefore, the current literature not only lacks descriptive, exploratory research on people working in pharmaceutical research and development laboratories in general, but also lacks research on people working in corporate continuous manufacturing laboratories. Concluding, the authors aim to close this gap by addressing the following research question in this research article:
RQ: How do people work in corporate pharmaceutical research and development laboratories for continuous manufacturing, what are their respective needs, and how can these needs be met?
2 Methodology
2.1 Contextual inquiry and content analysis
The nature of organizations as emergent open systems, subsisting through on-going interactions of the individuals who act within them, means that it is necessary to ‘view them as human activity systems’ (Checkland and Poulter, 2006). In this regard, people often fail to manage organizational change and innovation, because they are not able to articulate and describe the current as-is situation of processes, workflows, habits, and people’s attitudes in place.
Therefore, methodologies like requirements engineering, ethnography, as well as user research and user needs analysis as the rather technology-oriented social research methodologies, aim to describe and clarify these as-is situations to derive meaning and offer insights for optimization and management. The explorative nature of the qualitative research methodology offers opportunities to explore contextual dependencies, both for individuals and in groups, and to question assumptions that are taken for granted (Bednar and Welch, 2014). This study follows a qualitative user research methodology to explore workflows and derive user needs of people working in a research lab. While user requirements refer to potential system qualities that need to be met for some kind of user satisfaction, User needs refer both to the difference between users’ goals and the present condition, which is manifested by user problems and possibilities, and the context of use, which includes the characteristics of the intended use reflecting a users’ present tasks and environment (Lindgaard et al. 2006; Kujala et al., 2001). User needs also imply that researchers focus on the underlying needs of participants and try to infer these broader needs on the basis of what participants are saying during interviews. Thereby, the researcher deliberately separates the interviewee’s wishes (sometimes called “user wants”; Yi, 2018; Hartson and Pyla, 2019).
In order to build an understanding of how work in a modern pharma R&D laboratory is conducted and what the individuals working in this environment actually need to perform tasks and reach their goals, participating obser- vations with live interviews are conducted.
This applied research method is also known as Contextual Inquiry, which originates in ethnographic research traditions coming from anthropology (Plowman, 2003; Stanton et al., 2013; Ladner, 2014). In addition, a master-apprentice approach is applied in which the researcher takes on the role of an apprentice treating the interviewee as an expert in the current situation and tasks at hand (Downey et al., 2015). From a practitioner’s point of view, such a Contextual Inquiry can be considered an extremely well-prepared customer visit, site visit, or field visit (Goodman et al., 2012) to gather insights of users within the context of use.
The core of contextual inquiry is to conduct focused observation and having a conversation while a user is performing a task of interest. This includes gathering artifacts, taking field notes, while at the same time conversing with the user in an informal manner. This requires the researcher to be adequately prepared, travel to the workplace, follow the appropriate requirements for access, gain approvals for recording, and bring along all the necessary equipment. Having a plan to collect reliable data is necessary, especially in pharma laboratories, where schedules are quite busy and at times uninterruptible (Privitera, 2015; Werner and Kirsten, 2003). Analysis of contextual inquiries explore users’ experiences, aspirations, sense-making processes, and surface their tacit understandings of contextual dependencies in the context of their ‘problem space’ (Bednar and Welch, 2014; Privitera, 2015).
In the following sections the results of a contextual inquiry in a selected R&D chemistry laboratory focused on continuous manufacturing are presented. Thereby, the authors aim to describe the as-is situation and dependencies of actual lab work including the elicitation of user need. These insights shall build a body of knowledge to identify challenges and design implications for chemistry laboratory workplaces. Laboratory staff is interviewed during their daily work in the chemistry lab with a focus on workflows and used tools. Additionally, videos are recorded to optimize the work-flow analysis process, resulting in four hours and 47 minutes (287 mins) of video documentation. On this data basis, transcripts of the interviews are created containing key images of the video recordings to set up an ‘authentic’ set of qualitative data (Ladner, 2014). Then an in-detail content analysis is conducted, which is an extensive, transparent, stepwise interpretation process applied to the qualitative data gathered (Mayring 2000). Here, a researcher sorts the text snippets of transcripts into groups and assigns codes, e.g. values, roles, goals, tasks, mental model, behaviors, pain points, or mistakes (Goodman et al., 2012).
2.2 Sample
As the literature review illustrates, gaining access to corporate research facilities for conducting social research cannot be taken for granted. In addition, the goal of this study is to study not only people in a special research facility for continuous manufacturing, but people working within a big pharma company. Therefore, recruiting lab managers for this study is a challenge. While the recruitment process included lab managers from several of the world’s biggest pharmaceutical companies, only one company was interested to cooperate for the study investigating human factors for continuous manufacturing. In addition, the scope of the cooperation for this study was limited to conduct research in one laboratory. Also, the access to the laboratory was limited to one day, which corresponds to participating in one day shift of lab work in this laboratory.
All in all, five in-depth participating interviews are conducted in situ during one day shift and one 70 minutes expert interview with the respective lab manager to cover the bigger picture of the current organizational setup of the lab and its possible future direction. In detail, the set of participants contains a team leader for a group of technicians (called ‘Operations Team Lead’), who reported eight months of experience in this laboratory, one Continuous Processing Engineer (called ‘Engineer’) with one year of experience in this lab, and three Continuous Processing Technicians (called‘Technicians’) with many years of experience.
3 Findings
Regarding the question, how people work – and what they need to do so – in corporate pharmaceutical research and development laboratories for continuous manufacturing, the findings from the qualitative content analysis are summarized and presented in four steps. First, the laboratory setup is explained. Second, general observations are summarized describing the organizational setup. Third, the observed workflow for analytical method development is described. Fourth, responsibilities, needed information, and needed intercommunion and advocacy regarding the job roles of study participants are summarized. Finally, a summary of initially 96 elicited user needs (see appendix) is presented in four overarching categories of people’s needs during lab work.
3.1 Laboratory setup
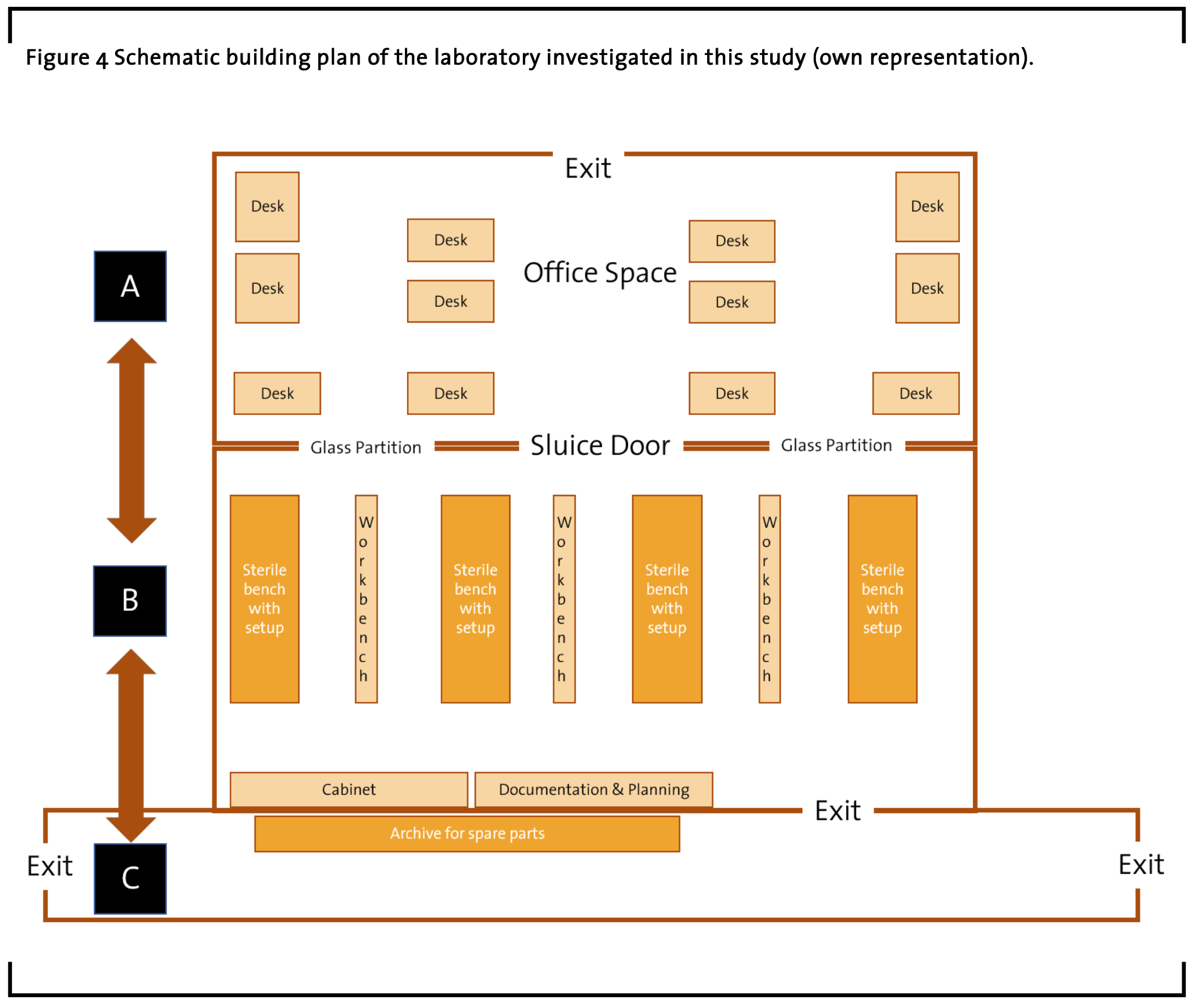
In order to understand the activities involved in working in a laboratory focusing on continuous manufacturing, the first step requires highlighting the layout of the equipment besides the processes of teamwork and communication, hardware and automation setup, and maintenance, as well as testing and data analysis. In the following section a rough scheme of the laboratory floor plan is presented, which does not include the actual setup of additional sinks beside sterile work benches or safety related equipment that is mandatory in such research facilities. Overall, the laboratory space can be divided in three main regions A, B, and C (see Figure 4).
Section A constitutes the office space where responsible scientists (chemists, pharmacists, engineers) meet, plan, analyze data, and manage their research projects that are conducted in the laboratory next door. This area is connected to the overall office space of the building.
The section B represents the actual research laboratory where chemical experiments are set up in big sterile work benches to test chemical recipes in different scale, ranging from very small, microscopic experiment setups in the beginning to medium sized setups occupying the complete space of a big sterile work bench in the end of a test series before a possible hand over to other departments responsible to scaling the setup to big production plants. Here the chemistry is tested using pressure controllers and computer software for recording data. The workflows deal with chemicals as well as specialized equipment. Some equipment and chemicals are stored in a cabinet directly located inside the laboratory.
But most of the equipment is placed in the hallway outside the laboratory, in section C, in a manner that separates the hardware archive while providing adequate safety. This hallway is connected to the suppliers input areas and other laboratories. Along with the above setup, safety is a key concern in the laboratory. Technicians and scientists working in the laboratory have to undergo mandatory safety training. Everyone entering the laboratory uses glasses, gloves and laboratory coats to ensure precaution during work hours. As mentioned above there are additional sinks and eyewash stations as well as steps outlined about what is to be done in case of an emergency. There are also emergency contact numbers and a phone available in the laboratory. All employees involved take the utmost care during the setup as well as during the testing process. Typically, there are considerable safety risks involved in this work. All handling of dangerous chemicals is done under the sterile work benches which contain a ventilated fume hood.
3.2 General observations
The overall goal of the investigated R&D chemistry lab is the continuous design and evaluation of chemical recipes for drug development on a small scale. Validated recipes are then scaled to a bigger development setup until they fit the needed outcome on the one hand and regulatory norms on the other hand. Usually, several recipes on diverse scale levels are tested side by side in one lab environment. The recipes are tested with specific setups under controlled conditions in fume cupboards. As sterile work benches have glass doors a lab worker can easily have a look at the current hardware, which is the experiment setup for testing a chemical recipe. The desks of the lab manager and responsible chemists are visibly placed in the glass office right next to the sterile experiment laboratory. This spatial setup enables ad hoc discussions between the operation teams, chemists, and lab management.
In the beginning of a shift, the team of engineers and lab technicians takes a look at the current project plan to clarify the current situation the last shift team ceased to work and in what condition the respective experiment setup is right now. Besides these daily check-in and check-out meetings, the operations teams meet with the chemists that are assigned to their project on a regular basis.
In general, the team differentiates three overall workflow phases: (1) Inventory Management, (2) Setup, and (3) Risk Management. First, technicians take a look at the current state of the hardware setup they are assigned to. They walk up to the sterile work bench they are assigned to, visually inspect the experiment setup and read the process diagram poster that is attached onto the front of a sterile workbench. The process diagram posters are prepared by the assigned chemist. The technicians look for planned changes to the experiment setup and then check for additional hardware components that are needed to rearrange the setup accordingly. Hardware components are stored in a separated inventory. Technicians then collect the needed components and iteratively change the setup according to the process dia- gram. During their lab work they are always in correspondence with the overall team and the assigned chemist or their lab manager to clarify information or discuss issues like e.g. how to document a specific abnormality or which component could be used alternatively in order to optimize the overall experiment setup. In general, everyone involved is required to document any changes they make, abnormalities as well as incidents they observe. Therefore, the lab management provide printout documentation templates called “proforma” on a regular basis. These printouts cover specific sub-processes like cleaning a specific part of an experiment setup and are stored in a specific cabinet inside the lab.
In addition, general paper lab notebooks are used for overall documentation, which are covered in red color. These lab notebooks are then placed upon the work benches opposite to the sterile work benches.
Besides these assets of pen and paper docu- mentation there are several laptops and desk- top computers installed on top of every line of work benches to provide access to company internal IT systems. These IT systems include for example: An Electronic Lab Notebook, Electronic Ordering Systems, and Equipment Booking Systems.
Currently problems occur at the intersection of several ongoing processes, that is, the synchronization of research design, management of employees, concrete project planning and resource management. This also includes the supply of expensive resources and equipment over long periods of time. Momentarily a central problem in the laboratory is the finding of specific equipment. This is especially difficult because equipment is shared amongst the different teams and there can be long time elapses during projects, e.g. it is possible that there are two years between the first and second use of a component part. Thus, the corresponding need for a systematically processed storage system and dynamic scheduling tools becomes stronger.
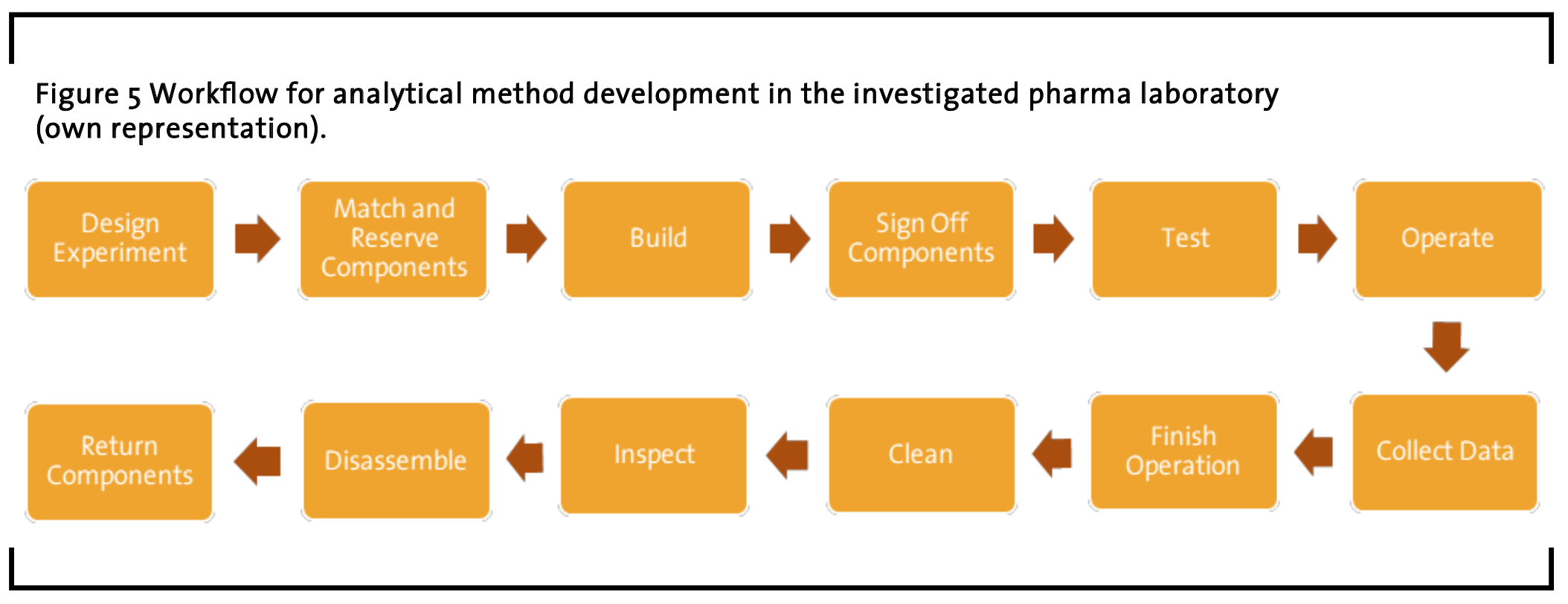
3.3 Workflow for analytical method development
In analytical method development people are looking to build appropriate testing platforms that are able to meet the needs of the process as well as provide a platform for consistent operation and data processing. The process starts off with a hypothesis of the mechanism for the chemical transformation. This results in a number of potential experimental designs that can be used to test this. An experimental design is always assessed against the equipment capabilities. There is a library of modelled equipment that is available to be used to create the test conditions needed to explore the mechanistic hypothesis of how a chemical recipe might be applied technically. Here, the equipment is matched against the proposed experimental requirements and the operational recipe developed. Modular automation is used to apply standard automation controls to the assembled platform. All aspects of the platform configuration are captured in the design process to ensure that full context information of the experimental design is documented.
These details include all factors that could impact the repeatability of the experimentation, which in flow includes all aspects of the wetted flow path, such as interconnecting pipework geometry, ambient conditions, equipment baseline characteristics. All the experimental design is held electronically in an electronic lab notebook, with appropriate experimentation context to allow for indexing.
Once built and tested (baseline standards) the platform will run a number of transient and steady state experiments to allow for efficient data collection with minimal material utilization. Operation of the platform, although automated, still relies on human oversight, and this is carried out through a number of dashboards that support both experimental and operation aspects.
Each experiment is monitored and data analyzed as close to real time as possible – dependent on the complexity of the model – to ensure that experimental requirements are met before moving to the next run. Where experimental data is not in line with the hypothesis, the operator is informed as a user response is required to move the process forward.
At the end of an experimental run, the system is automatically cleaned and inline sensors determine the end of the cleaning cycle. During the cleaning cycle the equipment performance is again baselined to determine any impacts from experimental processing. Then follows cleaning and an inspection is carried out, which is then followed by disassembly (further inspections at this time), and any observations made to the experimental procedure is documented. Components are then returned to stores and the electronic equipment tracking system is updated. The workflow comprises the following steps in the given order (see Figure 5):
(1) Design Experiment: Initially, experiments are being designed by scientists based on previously conducted experiments or prior knowledge. The outcome of this step are recipes and instrumentations, which are filed in some repository supporting versioning. The design phase includes an intensive negotiation between scientific and technical staff that aims to obtain an implementable experiment design. Specifically, a joined risk analysis is conducted and documented, which is not covered in the above figure.
(2) Match and Reserve: Components Given a recipe and instrumentation plan, requirements imposed by both are matched against specific physical components represented in a component inventory database. The outcome of this step is a bill of materials accounting for specific components that have been reserved for experimentation and that need to be collected. The reservation status of components is updated accordingly. If matching fails, experiments need to be redesigned or rescheduled. This step usually also includes collecting of components. Here, technicians search and collect components from physical inventories based on the bill of materials, which is embedded into a record containing all information relevant to the experiment. If component can actually be found, their status is updated accordingly. If components cannot be found because they are either not available or obviously broken, other components have to be found in another match-and-reserve components step.
(3) Build: Technicians assemble the experiment based on the mod-prep record. If components turn out to be incompatible, technicians have to go back and repeat the match-and-reserve components step.
(4) Sign-off Components: After successful build-up of the experiments, all utilized components are signed-off in the component inventory database and, thus, made unavailable and locked for further use. The usage status of components is updated accordingly.
(5) Test Experiment: Instrumentations are required to be tested prior to ordinary operation. In case this step fails, technicians have to revert to the match-and-reserve components step in order to remedy failures. Testing involves running a system with proxy solvents as well as with the target agents.
(6) Operate: Successfully tested experiment instrumentations can be operated. Data obtained during operation is stored in locally or remotely in respective data silos (databases). Operation of experiments is conducted according to a manual called Process Guide.
(7) Collect Data: Experiments that have been run successfully for the desired amount of time require the collection of data that is not automatically transmitted to back-end data storage.
(8) Finish Operation: Technicians shut down experiment operation.
(9) Clean Components: After experimentation, equipment must be cleaned superficially by technicians. The cleaning state is adapted accordingly.
(10) Inspect: Technicians inspect components after cleaning of experimental setups in order to check their functionality. Component’s functional statuses are updated accordingly.
(11) Disassemble: Technicians disassemble the experimentation setup and prepare individual components for their return to storage.
(12) Return Components: Individual components are returned to the component inventory and their usage status is updated accordingly.
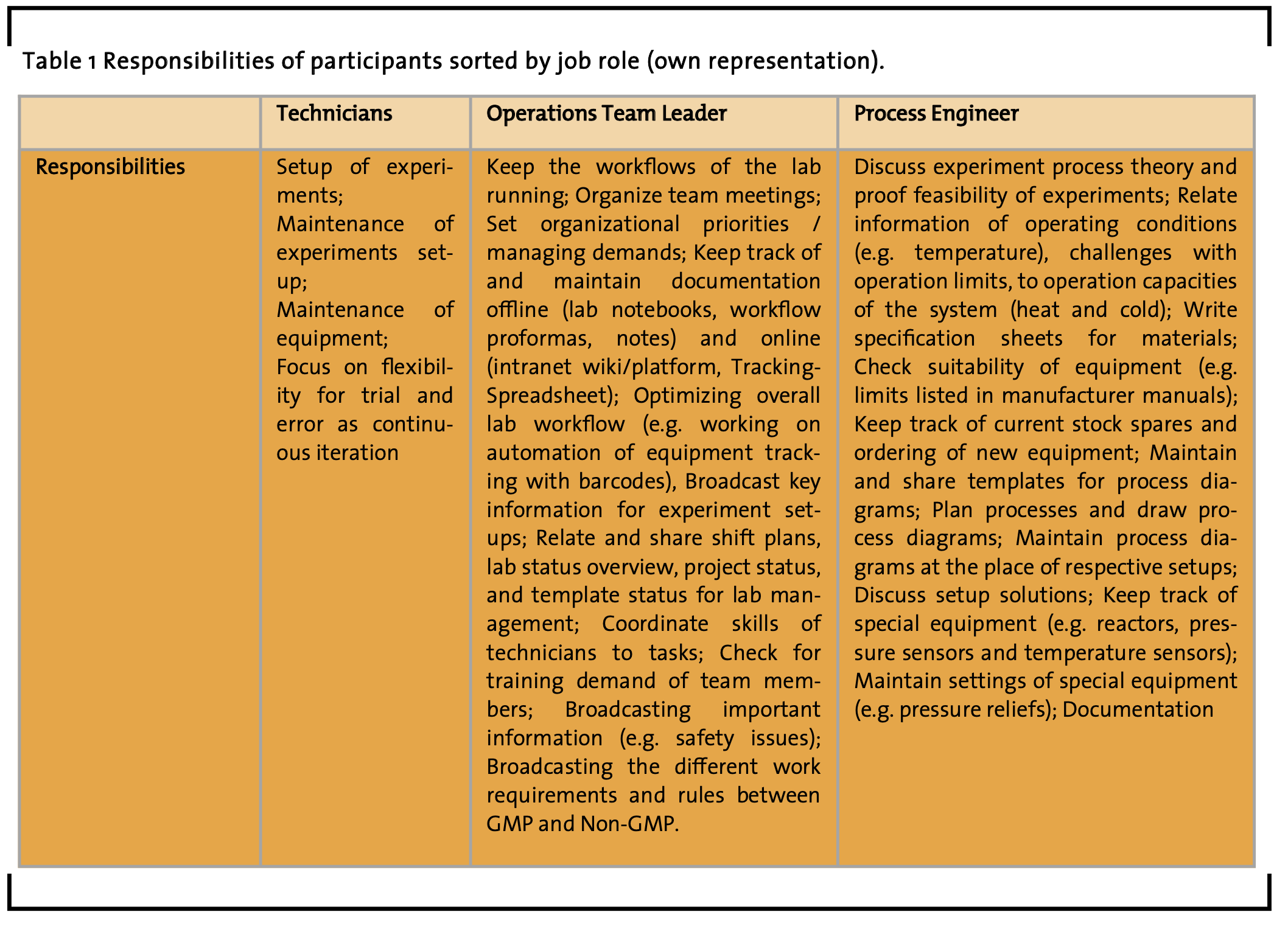
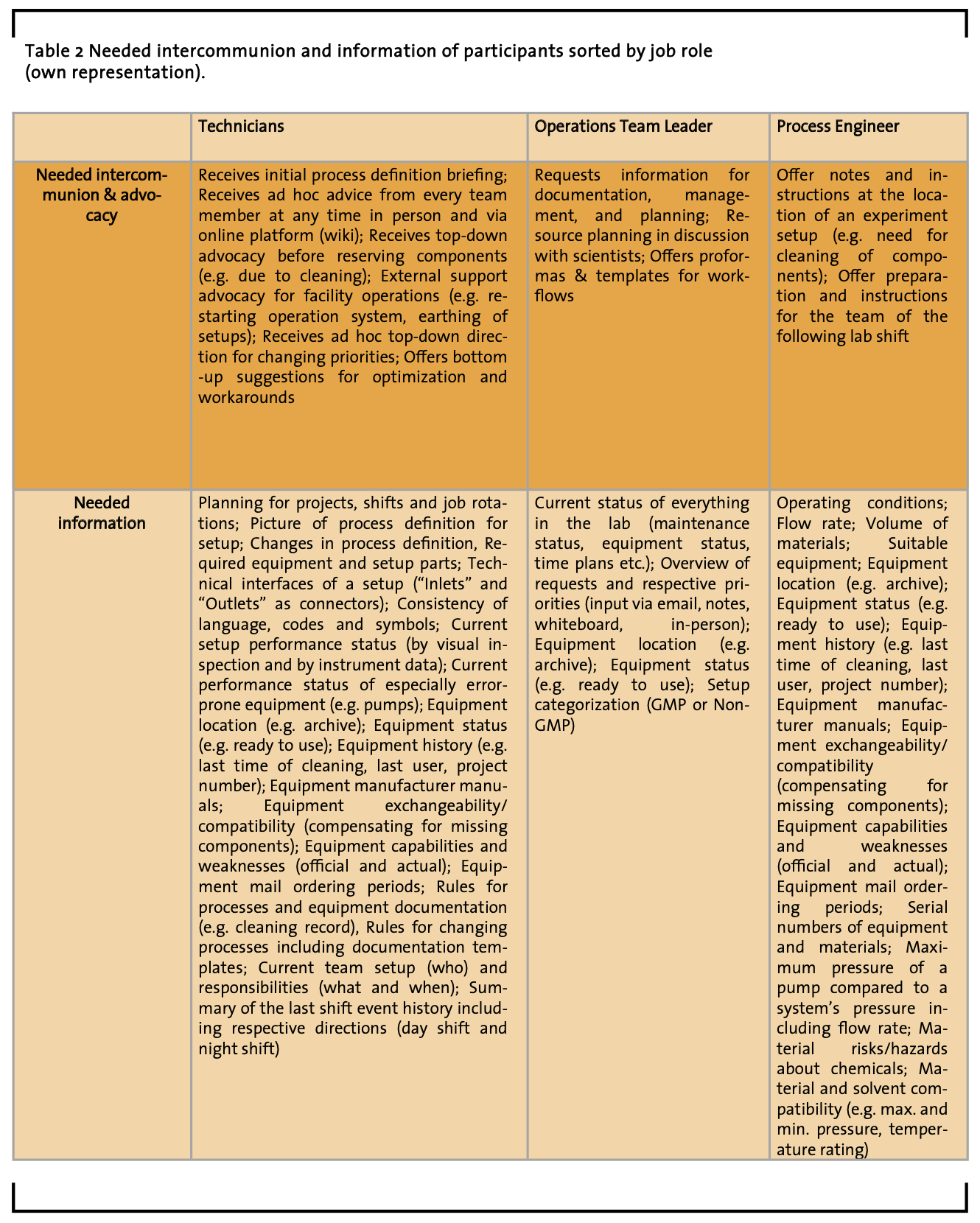
3.4 User roles
This section contains results that summarize (a) responsibilities (see table 1), (b) needed information (see table 2), and (c) needed intercommunion and advocacy (see table 2) regarding the job roles of study participants. The authors consider this kind of job role descriptions a foundation for the creation of people’s role profiles that may help other researchers, designers, software developers, engineers, and manufacturers to initially specify and discuss the user requirements of respective workflows, lab instruments, and lab software. Such summarizing role descriptions are considered highly important in human-centered design methodology, because they offer a set of information clarifying which type of user might need what information in which situation. Therefore, these role descriptions are referred to as ‘user roles’, sometimes also called ‘user profiles’ or ‘personas’.
3.5 User needs elicitation
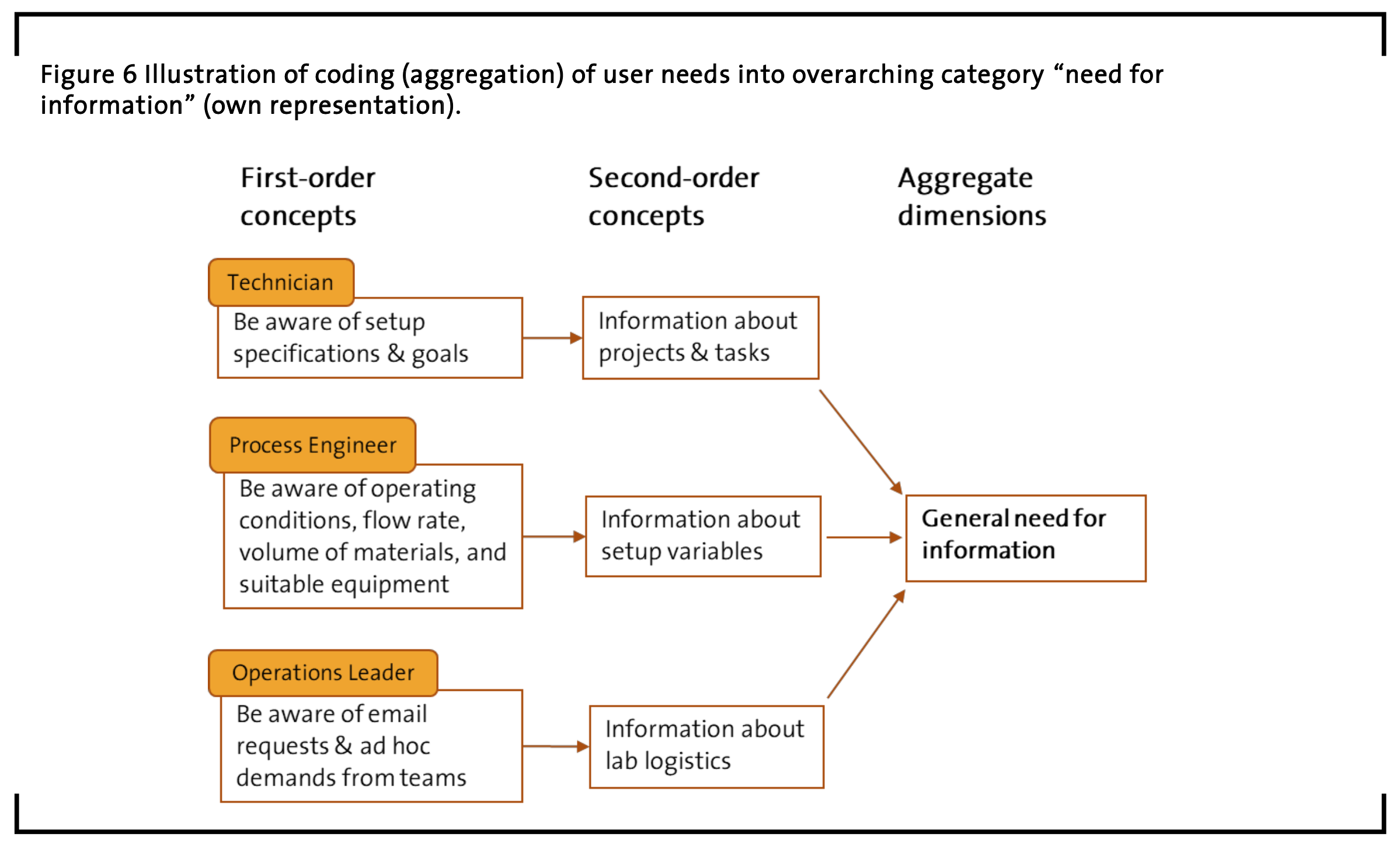
Another result of the conducted content analysis is an extensive list of 96 elicited user needs referring to things, information, knowledge, or communication the observed and interviewed participants need in their work environment to perform tasks and reach their respective goals. The complete list can be found in in the appendix. It contains redundancies as different users share the same tools and lab space and report similar things respectively. Therefore, it represents the complex mesh of everyday lab work that needs to be considered when discussing lab management, lab processes, workflow optimization, or the potential development of hardware and software for chemistry laboratories. But for reasons of readability presenting the complete list would be beyond the scope of this paper. However, the authors consider the following summary to be sufficient for understanding the main user needs and their interdependencies that occur in the flow of the studied lab work.
Therefore, the complete list is considered ‘first-order’ content regarding user needs inter- pretation. ‘First-order’ user need statements refer to concrete tasks, goals, or concepts like “checking pressure values”. The complete list is broadly grouped by the mentioned job roles (scientist, lab technician, operations team leader, engineer) and – where possible – by application context (design, setup, communication, documentation), while the actual user need statements all follow a consistent syntax containing (a) which job role a user has (b) what a user needs (c) as well as a contextual reason why a user needs it.
On this basis, summarizing categories or clusters of user needs are generated resulting in two further levels of interpretation and respective coding: ‘Second-order’ concepts that generalize the first-order statements, and a final ‘third-order’ level, here called ‘aggregate dimensions’, which represents the most general interpretation of groups of statements across job roles. The following section highlights some examples for this qualitative data analysis and stepwise interpretation process.
General Need for Information
The observations and interviews from the contextual inquiry show a “general need for information”. This category contains user need items that reference needed types of information (see examples in Figure 6).
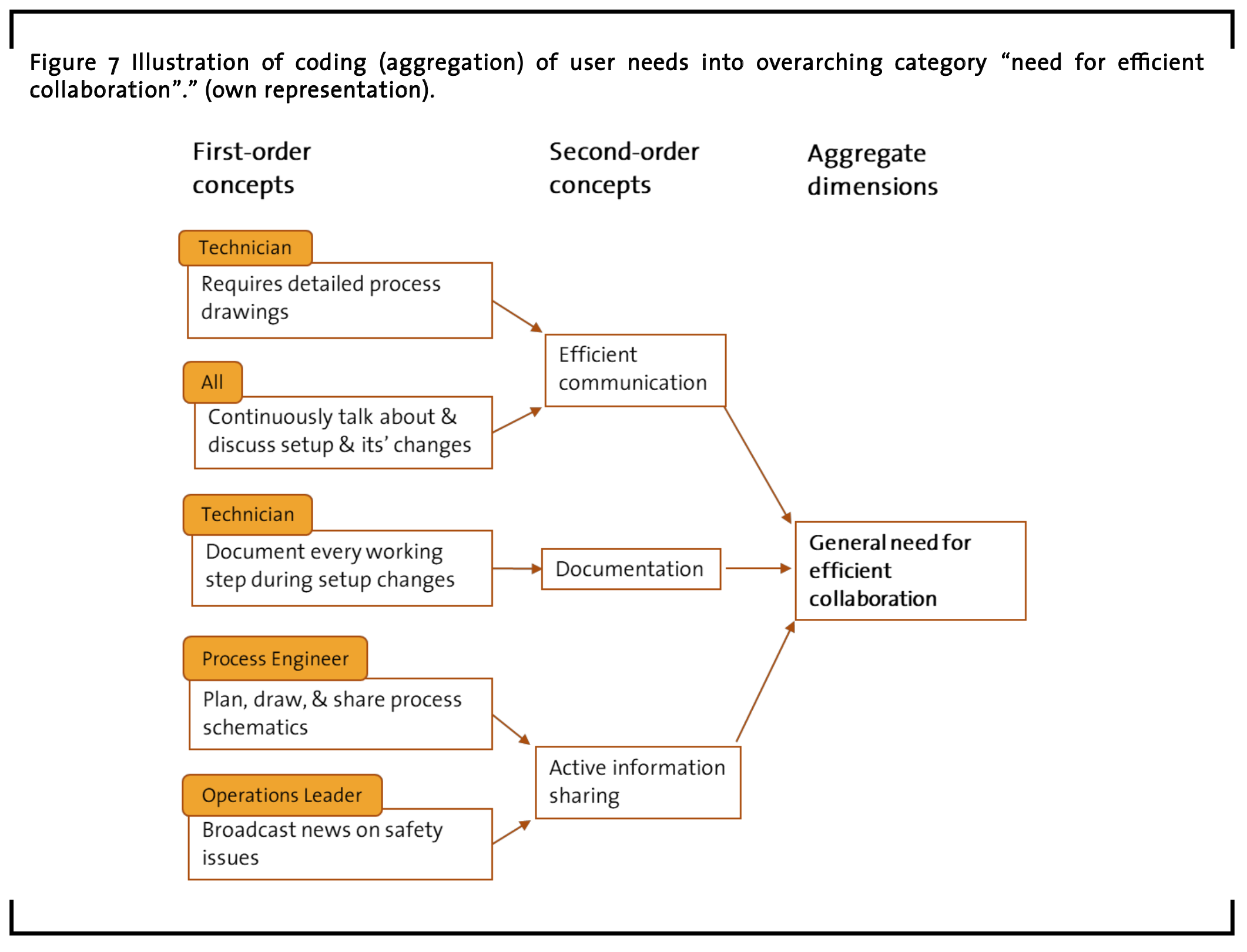
General Need for Efficient Collaboration Another overarching category that contains multiple user need statements is named “general need for efficient collaboration”. Here, user need items that address the need for efficient communication, documentation, and active information sharing are summarized (see examples in Figure 7).
General Need for Control
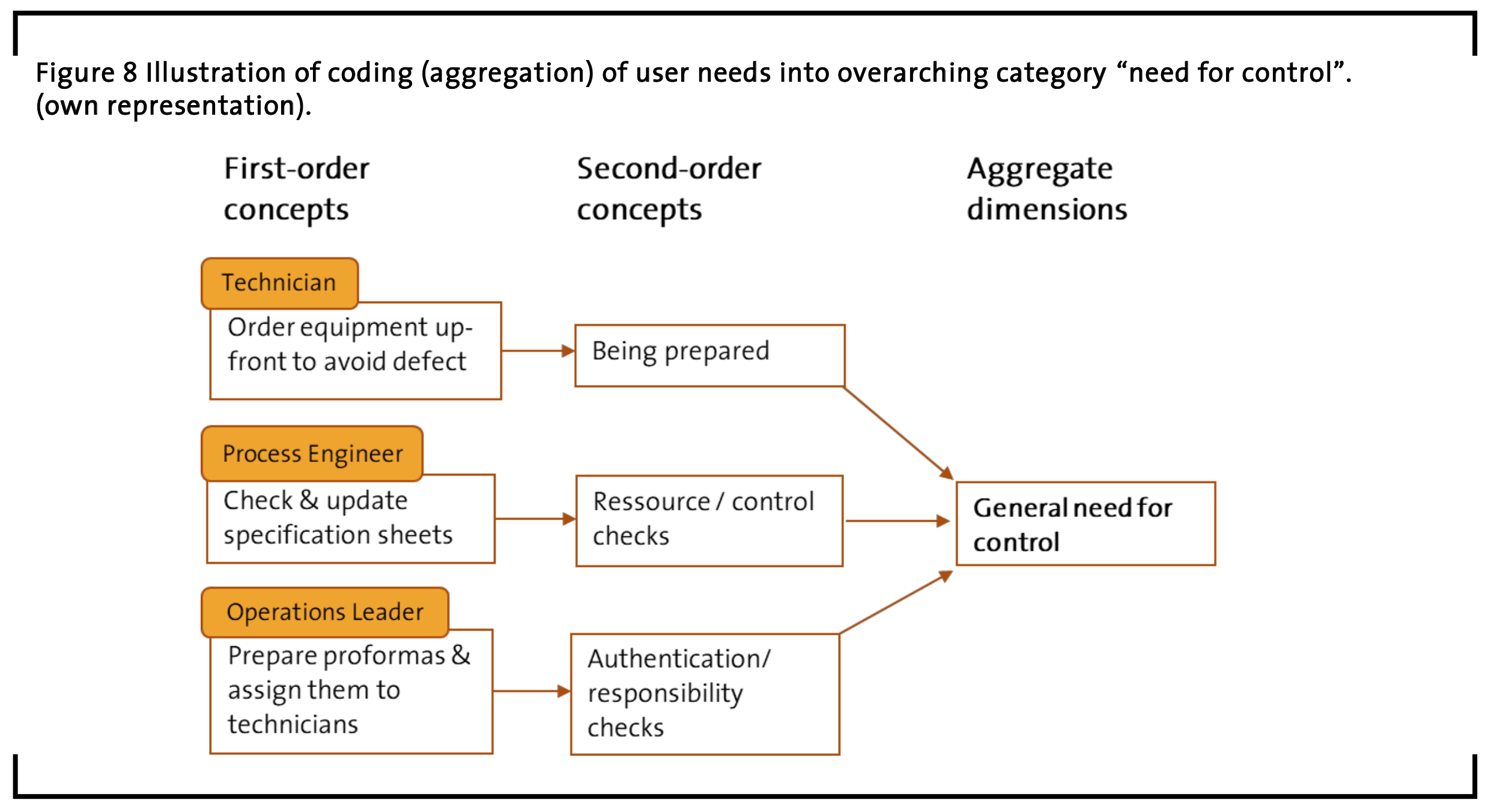
The third category that contains multiple user need statements is called “general need for control”. This category addresses user need items regarding preparation or being prepared, reassurance or control checks, as well as authentication or responsibility checks (see examples in Figure 8).
General Need for Flexibility
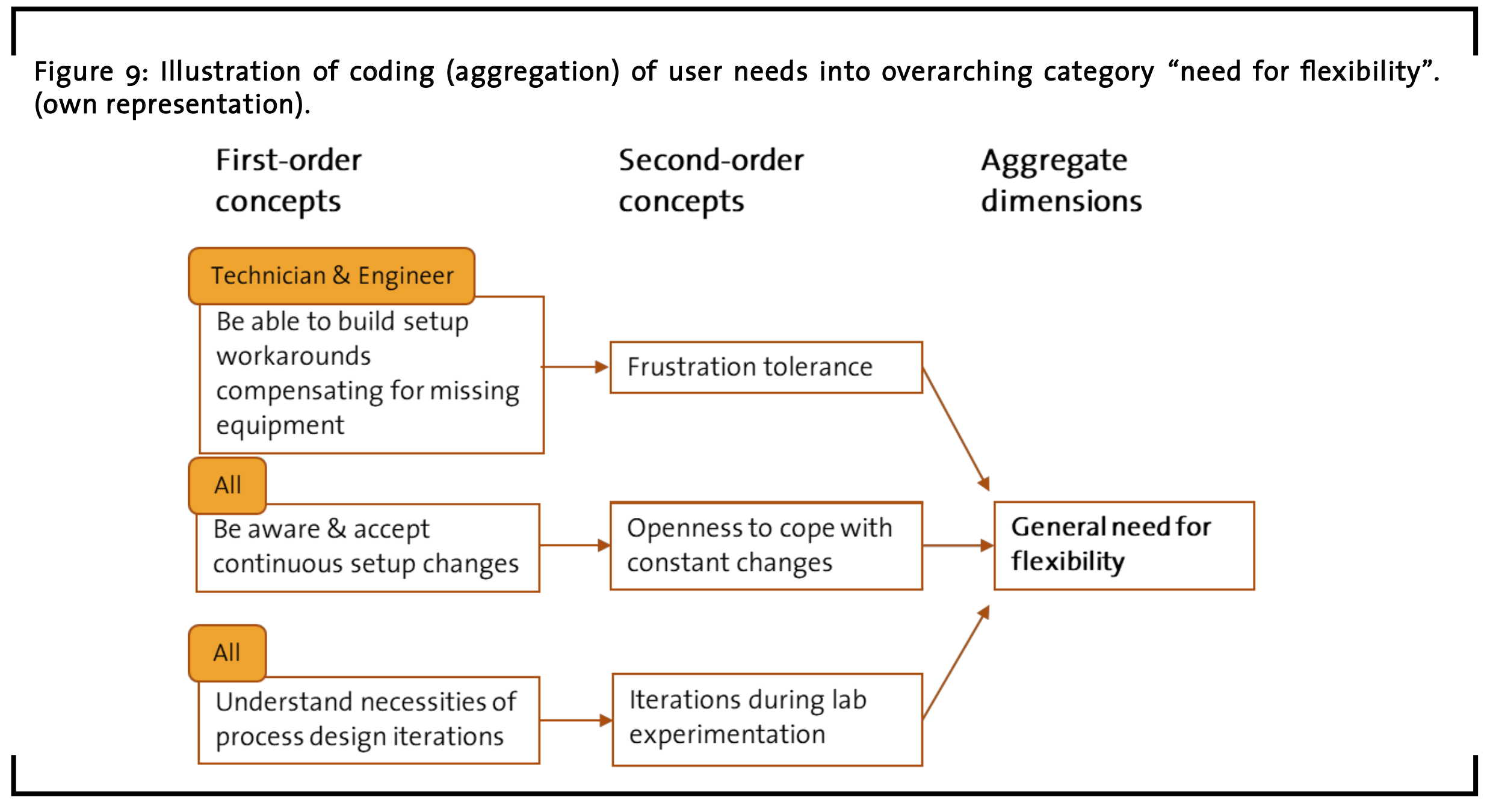
The last summarizing category is called “general need for flexibility”. Here, user need items that address issues regarding frustration tolerance and openness to cope with constant changes as well as iterations during the lab experimentation are collected (see examples in Figure 9).
4 Discussion
Employees working in pharmaceutical R&D for drug discovery and manufacturing prepara- tion face diverse challenges in all aspects of everyday laboratory work. Against this background, this study addresses the question of how people work in corporate pharmaceutical research and development laboratories for continuous manufacturing, what their needs are, and how these needs could be met.
The results of the current qualitative study show their extensive and diverse set of interrelated (user) needs from their employee’s point of view. Typically, these needs are supposed to be supported by e.g. rule sets like good manufacturing practice (GMP), safety regulations, and trainings. Looking at the literature, the importance of these ‘human factors’ has been emphasized.
For example, while Konstantinos and colleagues (2011) on the one hand mention future trends in pharmaceutical manufacturing and in industrial manufacturing as a whole, which reinforce the need of a stronger utilization of human factors aspects, as many issues of concern can be avoided or their effect minimized. On the other hand, their results show that compliance with the legislation regarding GMP regulations still seems to be the main requirement for pharmaceutical production, while human factors are not explicitly emphasized.
However, in the context of the results of this study these traditional rule sets and top-down management activities do not support all aspects of the employee’s experience. Overall, aspects of team culture and communication seem to be as important as these traditional approaches for safety and risk management.
Therefore, based on the results of this study several implications are discussed as general findings.
Finding 1: Digitization efforts should prioriti- ze communication and collaboration over features.
The results of this study also show that the lab workers struggle with the currently not consistent digitization efforts regarding documentation and organization of materials, solvents, and setup components. Digital tools like an Electronic Lab Notebook (ELN) or Wiki as knowledge base are already in place in this research laboratory, but are used to a less degree, because they either do have technical limitations or are limited in provided features – so they do provide some benefits, however, are not comprehensively deployed and thereby do not fit the user needs for constant and consistent communication during collaboration identified in the inquiry. Therefore, the support of effective collaboration is identified as a major challenge for the digitization of lab work, that is the continuous transition between both the analog and the digital world. For example, there is no consistent electronic documentation system in place and just specific workflows take advantage of specific features of such tools. So, on the one hand it is still common practice to use paper-based lab notebooks. On the other hand, there are a lot of ELNs available on the market. This, however, seems to be common practice in the majority of laboratories and evidence suggests that whilst scientists willingly make use of generic software for note taking and documentation, spreadsheets, general office software, as well as special scientific software to aid their work, current ELNs are lacking the required functionality to meet the needs of the researchers (Kanza et al., 2017). For example, most of available ELNs offer basic cloud-based text functionality like Microsoft Word while many of them fail to offer basic not to mention convenient capabilities of data sharing features. Based on this study’s results it is argued that such tools like ELNs should offer data and knowledge sharing as a main feature next to documentation for teams working in research laboratories. Data sharing thereby includes features like transparent versioning of documents and visualization of data wherever possible in order to enable respective knowledge sharing and collaboration. Goal should be to not simply replace existing solutions, e.g. replacing Microsoft Word or Excel by a process development tool not just for the features or to digitize an analogue process, but more so if the latter is enabling better collaboration by fostering the right organizational climate that persuades people to create, reveal, share and use knowledge (Davenport et al., 1998; O’Dell and Grayson, 1998; Mariani, 2002).
Here, again, the fact can be highlighted that in the development of new lab workflows or refinement of current workflows there are pro- cess plans and instrumentation blueprints that are communicated as diagrams, while the collaboration happening with these diagrams is not yet digitized consistently. But this exactly might be one issue to enable better communi- cation and thereby avoid mistakes and work- flow errors. In this regard, the current investigation shows challenges in communication, e.g. by means of ad-hoc documentation with print-outs and lab notebooks in correspondence with partially available digital documentation and knowledge sharing possibilities. For example, one technical solution addressing this finding may be to include interconnected large display covering live data, which could replace the mentioned paper-based setup diagrams that are currently pinned on the front of sterile work benches. Thereby, research laboratories might benefit from the logistics of so-called ‘control rooms’ (Lischke et al., 2018).
Finding 2: Learning is considered a shared responsibility to foster an agile work culture for efficient in-team knowledge management.
In order for the lab team to collaborate efficiently there needs to be constant dialogue of team members fostering knowledge sharing and thereby collaborative learning. This finding relates to the general best practices in software engineering that follow a lean project management approach called ‘agile’ (Cardozo et al., 2010). Agile is a project management system where so-called ‘sprints’ or iterations are utilized to complete allocated tasks and assignments. Sprints or iterations are short spans of time, usually two weeks, where a team meets up and discusses the cycle of a project. In these meetings, tasks are assigned according to a proper timeline. Also, team work plays a significant role in the success of a project and there are frequent interactions between the client and the developers. Although there seems to be a common understanding, that agile development methods conflict with the generally accepted software development methods within the pharmaceutical industry (Hajou et al., 2014), it can be argued that the scientific lab work in the investigated pharmaceutical R&D laboratory shares some key dynamics to agile team work, following an overall mindset of adapting to change including trial and error for successful and constantly evolving collaboration. One key aspect of agile methodology is constant communication or focusing on relationships to foster team work and knowledge sharing. This finding is supported by the results from Mariani (2002) who describes the working culture in a very successful research laboratory in a way that resembles the ideal type of agile development methodology (Beck et al., 2001). In addition, some application-oriented publications argue that agile methodology is showing itself a promising way of working and moving beyond software development projects and soon will find its place in other industries like pharmaceuticals (Alaedini et al., 2014).
Overall, the results of this study suggest to lean more towards theories of human factors and education in lab management to foster this kind of sociocultural, organizational way of learning. For example, Boreham and Morgan(2004) propose a sociocultural model which identifies ‘dialogue’ as the fundamental process by which organizations learn, and relational practices as the social structure which embeds the dialogue and makes it sustainable in a potentially conflictual environment. They define a pedagogy of organizational learning in terms of participation that builds on relationships between employees in addition to the traditional focus on autonomy of individuals. This argument is also supported by Konstantinos and colleagues (2011) who state that lab workers need to interact as to form social networks, interactions and teams that can facilitate group identification and lead to more smooth coordination and collective action, while the parameter of auditing can be an issue among the workers and again prohibit the teamwork (Hutchinson and Zain, 2009). In addition, these authors generalize such issues stating that in many organizations, a major cultural shift is required to change employee attitudes and behavior so that they willingly and consistently share their knowledge and insights and thus help management and control process. Another study supporting this argument showed that in order to be successful in pharmaceutical R&D teams depend on the capacity to perform a large number of experiments and the capacity to rapidly modify or adjust production programs (Mariani, 2002). Here, the call for flexibility, continuous learning and teaching of team members, coupled with an increased rate of experimentation challenging strict regulations including confidentiality is especially interesting.
Finding 3: Incorporating user research prac- tices is important to uncover underlying social systems as determinants for the success of socio-technological systems.
One goal of this in-depth exploratory user study is to foster fundamental and interdisciplinary debates about the design and implementation of applications based on novel hardware and software products in laboratory environments. Such debates are considered very important since the conceptualizations of managers, engineers, developers, and designers are often based on initial hypotheses concerning the collaborative relationship between their technological product and the potential end-users, while these collaborative relationships are often neither made explicit nor are these assumed relationships verified in the field (Sharit, 2006). Furthermore, the conduct of validation and verification studies during product development and after product release is usually limited to the technical side of implementations, thus results may be informative but hardly about a combined view of social and technical aspects. On the other hand, the potential to succeed or to fail of a system, once it has been brought to the field, is strongly determined by the adaptation of both social and technical aspects (Dekker, 2005). In this regard, the reward structure of organizations usually emphasizes on rapid completion of projects and the insula- tion of engineers to give less consideration to factors related to ease of operation and even safety (Perrow, 1983). Here, the results of this study show the interdependencies between all individual employees working in the lab, their need and constant effort to initiate and maintain an efficient mode of communication and collaboration as a foundation for the technical side of chemical research and recipe development.
The most striking yet straightforward example for this interdependency of responsibilities and respective communication routines is found in the collaboration around the already mentioned process design drawings. Here, a scientist creates and proposes a process drawing which follows his current experimental hypothesis in order to discuss the technical feasibility of a respective process setup in the lab. The process engineers and technicians on the other hand rely on this process drawing in order to understand what they are supposed to do next, while also anticipating the broader research idea of the scientist. In theory, this collaboration seems like a top-down approach of research design and management, but the results show that it is rather a constant debate or iterative collaboration for setup refinement to find technical solutions as a team, which need to be feasible yet do not compromise the scientific hypothesis. Surprisingly, interview partners also mentioned that technicians sometimes have more in-depth knowledge about equipment and instrumentations than their highly skilled scientist and engineering colleagues. One interview partner even mentioned that occasionally scientists first ask technicians to propose a setup instrumentation to iterate on a recipe experiment or that a recipe and its experimentation setup is changed, because of a proposed technical solution from a technician. Thereby, it can be concluded that the scientists include bottom-up experimentation in their research rationale. This statement also supports the finding regarding work culture in this lab to follow a mode of constant debate that tries to contain knowledge transfer in teams top-down as well as bottom-up.
In the end, the importance for lab management to identify and clarify such aspects regarding the flow of information for effective knowledge sharing in teams working in such fast-paced and ever-changing work environments can be inferred. Hence, team leaders or lab managers should know and communicate which team members need to know what information in what kind of form and when in which point in time from whom to what effect. This finding goes along with the research regarding the theoretical construct of ‘work process knowledge’ from Fischer and Röben (2002a), which highlights a special type of shared knowledge that team members working in organizations need to develop and continuously expand in order to anticipate the organizational requirements and the needs of their colleagues. Thereby, it can be concluded that it could be helpful and might become important for pharmaceutical companies in the future to incorporate such kind of user research or human-centered design practices to uncover such underlying individual needs and social interdependencies as determinants for the success of the socio-technological system that is the chemistry laboratory for research and development.
Generally speaking, it is suggested to research and analyze cultural and social aspects of workflows in special environments like laboratories before designing and developing digital solutions on the road to paperless work (Thimbleby, 2019). In addition, it is argued to respect and include bottom-up processes of collaboration that are based on experiential knowledge. Here, the increased cost of repetition (trial and error), e.g. by failed measurement, has to be balanced against the contribution to the development of individual competence and quality assurance within an organization (Fischer and Röben, 2002a).
This ‘cultural approach’ to lab knowledge management could also help organizations to specify opportunities and benefits regarding the adoption of new tools and solutions, which is often a challenge, because the benefits of outdated yet established tools and solutions usually predominate the adoption costs of new tools and solutions that, however, may offer more benefits in the long run (Rogers, 2003; Wang et al., 2010). For example, Schmitt (2019) investigates the challenges regarding the adoption of B2B electronic marketplaces in the chemical industry and argues for a top-down approval approach for introducing new solutions through managers, because on the one hand operative individuals in general show the tendency to stick to familiar processes and on the other hand there is often a lack of incentives for employees to take up the challenge of new digital solutions bottom-up. However, as mentioned above, decision making for such top-down introduction of new solutions could be informed by bottom-up processes of cultural analysis and knowledge management.
This is considered highly important, because beyond the study of lab work, these circumstances would appear to shift the attribution of user errors from engineers, developers, and designers to management in the development of laboratory hardware and software products. Following Reason (1990) and Sharit (2006), such organizational ‘management errors’ represent types of latent errors that are responsible for creating preconditions of user errors in the end. Finally, this misaligned error attribution problem created by poor engineering, development, design, or management policies traditionally have been duped on training departments to compensate for these problems (Sharit, 2006). Respectively, it is argued that one major deficiency in these development processes may be the hubris of decision makers underestimating the human factors in their planning, as well as the inability of employees and management to appreciate human fallibility by failing to take into account relevant information. Overall, the step of developing a Human Factors Engineering strategy or User Experience strategy or Human-Centered Design strategy is either not completed or it is accomplished by non-human factors personnel, which can lead to poor results (Privitera, 2019). In this regard, Privitera (2019, p. 28) explicitly argues:
“The reasons for this are varied; however anecdotal evidence suggests that manufacturers often believe only a minimum of Human Factors input is required based upon what they believe to be the agency requirements. Manufacturers then provide a rationalization that doing more is not necessary because they, in fact, “know their users”. In instances where manufacturers lack Human Factors analysis procedure, they may rely on hearsay without an optimized or documented approach. This becomes problematic in developing the required Human Factors submission documentation for agency review. Lastly, personnel without additional education in Human Factors may not ask the right questions necessary to determine appropriate human factors strategies required to optimize the device interface. Thus, developing a Human Factors strategy at the onset of a device program assures that all User Needs are met and that the manufacturer has a firm commitment to quality from the user’s viewpoint.”
Therefore, the notion of Ulbrich and Aggarwal (2019) is supported that companies will need “translators-specialists” – the aforementioned user researchers or human factors engineers – who are able to understand the functionality desired by e.g. lab workers and translate it into technical requirements that can be understood and processed by the IT staff. In this spirit, this exact concern is given serious consideration in human-centered design practices (Nielsen, 1995; Sharit, 2006; ISO/IEC 13407) and this study represents a source of insight for user needs in the context of laboratory workflows in general.
Limitations of this study
Nevertheless, the aforementioned results and findings need to be reflected in context of the methodological limitations of this study. These limitations include a lack of prior research studies on the specific topic of lab work in continuous manufacturing labs of big pharmaceutical companies.
Further research might also apply mixed-methods research designs combining not only observation and interviews, but also surveys or longitudinal data collection approaches like diary methods for continuous experience sampling. In addition, reliability of qualitative data is always an issue due to the methods for data collection relying on self-report data only. Here, it needs to be clarified that this study represents a first step in exploring the field of corporate lab work in continuous manufacturing and that more research is needed to generate more reliable data and respective insights. However, the most prominent limitation is the small sample size due to limited access to pharmaceutical companies as well as people working in these companies. Here, different modes and contract models of collaboration for co-innovation could be explored to get more access to corporate research facilities (Schneiderman, 2016), e.g. by supporting and fostering open innovation initiatives (Hunter and Stephens, 2010; Bianchi et al., 2011; Schuhmacher et al, 2013) between social researchers and corporations.
5 Conclusion
Scientists, engineers, and lab workers developing chemical solutions in big pharmaceutical companies construct, engineer, monitor, and manage complex processes. Staff members have to follow strict regulations while making system-relevant and sometimes safety-critical decisions. Thereby they are supported by a large amount of information in form of lab notebooks, drawings, or diverse format templates for documenting specific processes and incidents. This study provides an in-depth view into common work practices and aims to identify challenges for workflows, processes, decisions, and actions in a chemistry research laboratory for analytical method development in continuous manufacturing.
The findings show what people in this laboratory need to work together and communicate effectively. On top, in comparison to current psychological and social studies in the field of laboratory work, e.g. from the human factors community, the results show that there is a fundamental difference in research design focusing on human needs instead of human error. Thereby, the study follows the paradigm of positive psychology (Seligman and Czikzentmihalyi, 2000) instead of a paradigm of economic and mechanistic efficiency or technical functionality that typically considerers humans as a source of risk and technical errors, hence, the traditional goal of such research initiatives is to optimize the human-machine system (sometimes even called ‘co-operation’) for reducing the potential for human error. For example, in comparison to these studies which offer the overall identification and listing of risk factors like “stress” or “time pressure”, this current study investigates and identifies the constitutional employee needs as user needs in situ that lead to such psychological and social phenomena. Hereby, this study is in line with the overall goal of human-centered design including the user-research-led product development practice of user experience design, which are both fundamentally focused on human needs and their relationship towards environments, systems, applications, and products. By providing an extensive list of 96 user needs (see appendix) and detailed descriptions of user roles this study aims to encourage a debate about hu- man-centered design in pharmaceutical manufacturing.
Finally, the findings support the notion of Ulbrich and Aggarwal (2019) that it is vital for chemical companies to incorporate employees in transformation processes, e.g. when implementing cloud and analytics solutions that are centered on the users, who are in this case scientists, engineers and lab technicians. In this regard, the study aims to fill a gap in discussions around enabling future laboratory processes. Here, typically project stakeholders are not able to describe the current as-is situation of processes, workflows, habits, and people’s attitudes in place, which is why the authors hope that this study also contributes to the overall discourse of change management and innovation in laboratory environments.
References
Ala, M., and Bagot, G. (1994): Ergonomics in the Laboratory Environment, Nursing Manage- ment, 25 (7), pp. 50–52.
Alaedini, P., Ozbas, B., and Akdemir, F. (2014): Agile Drug Development, Lessons from the Software Industry, Why pharma needs to move from a “waterfall” to a “scrum” model, availab- le at: https://www.contractpharma.com/ issues/2014-10-01/view_features/agile-drug- development-lessons-from-the-software- industry/, accessed 2 January 2020.
Allotrope Foundation (2018): Workflow Dia- gram, available at: https://www.allotrope.org/ resources, accessed 10 November 2019.
Amirebrahimi, S. (2015): Moments of Dis- juncture, the Value of Corporate Ethnography in the Research Industrial Complex, Ethnographic Praxis in Industry Conference Proceedings, 2015, pp. 13–23.
Aromaa, S., Väätänen, A., Hakkarainen, M., and Kaasinen, E. (2018): User Experience and User Acceptance of an Augmented Reality Ba- sed Knowledge-Sharing Solution in Industrial Maintenance Work, in: Advances in Usability and User Experience Proceedings of the AHFE International Conference on Usability and User Experience, 2017, Springer International Publishing.
Atkinson, P. (2015): For Ethnography, London, Sage.
Atkinson, P., Delamont, S., and Housley W. (2008): Contours of Culture, Complex Ethnography and the Ethnography of Complexity, Plymouth, AltaMira Press.
Badke-Schaub P., Hofinger G., Lauche K. (2008): Human Factors, Springer, Berlin.
Bahl, Anke; Dietzen, Agnes (2019): Work- based Learning as a Pathway to Competence- based Education. A UNEVOC Network Contribution, Federal Institute for Vocational Education and Training, Bonn.
Barley, S. R. (1996): Technicians in the Work- place, Ethnographic Evidence for Bringing Work into Organizational Studies, Administrative Science Quarterly, 41 (3), pp. 404–441.
Barley, S. R., Bechky, B. A., and Nelsen, B. J. (2016): What Do Technicians Mean When They Talk about Professionalism? An Ethnography of Speaking, Research in the Sociology of Orga- nizations, 47, pp. 125-160.
Beck, K., Beedle, M., van Bennekum, A., Cockburn, A., Cunningham W., Fowler, M., Grenning, J., Highsmith, J., Hunt, A., Jeffries, R., Kern, J., Marick, B., Martin, R., Mellor, S., Schwaber, K., Sutherland, J., Thomas, D. (2001): Manifesto for Agile Software Development, available at: http://agilemanifesto.org/, accessed 15 January 2020.
Bednar, P., and Welch, C. E. (2014): Contextual inquiry and socio-technical practice, Kybernetes, 43 (November), pp. 1310–1318.
Bianchi, M., Cavaliere, A., Chiaroni, D., Fratti- ni, F., Chiesa, V. (2011): Organizational modes for Open Innovation in the bio-pharmaceutical industry, An exploratory analysis, Technovation, 31 (1), pp. 22-33.
Bonini, P., Plebani, M., Ceriotti, F., and Rub- boli, F. (2002): Errors in Laboratory Medicine, Clinical Chemistry, 48 (5), pp. 691–698.
Boreham, N. (2002): Work process knowledge in technological and organizational development, in: Boreham, N., Fischer, M., and Samurcay, R. (ed.), Work Process Knowledge, London, Routledge, pp. 1-15.
Boreham, N., and Fischer, M. (2009): The Mutual Shaping of Work, Vocational Competence and Work Process Knowledge, in: R. Maclean and D. N. Wilson (ed.), International Handbook of Education for the Changing World of Work, Springer, Bonn, pp. 1593–1610.
Boreham, N., and Morgan, C. (2004): A Soci- ocultural Analysis of Organisational Learning. Oxford Review of Education, 30 (3), pp. 307–325.
Boreham, N., Fischer, M., and Samurcay, R. (2002): Work Process Knowledge, London, Routledge.
Boreham, N., Nyhan, B., and M., F. (2004): European Perspectives on Learning at Work, The Acquisition of Work Process Knowledge, Office for Official Publications for the European Communities, Luxembourg.
Bountra, C., Lee, W. H., and Lezaun, J. (2017): A New Pharmaceutical Commons. Transforming Drug Discovery, Oxford Martin Policy Paper, Oxford.
Cardozo, E., Neto, J., Barza, A., Franca, A., da Silva, F. (2010): Scrum and productivity in soft- ware projects, A systematic literature review, 14th International Conference on Evaluation and Assessment in Software Engineering (EASE), 2010, Keele University, UK, pp. 131-134.
Checkland, P. and Poulter, J. (2006): Learning for Action, A short definitive account of soft systems methodology, and its use for practitioners, teachers, and students, Wiley, Hoboken, New Jersey.
Ciommer, B. (1996): Gegenwart und Zukunft der Analytik, LABOR Trend, pp. 10.
Darrouzet, C., Wild, H., Wilkinson, S. (2009): Participatory Ethnography at Work, Practicing in the Puzzle Palaces of a Large, Complex Healthcare Organization, in: M. Cefkin (ed.), Ethnography and the corporate encounter, Reflections on Research in and of Corporations, Berghahn Books, pp. 61-94.
Davenport, T.H., De Long, D.W. and Beers, M.C. (1998): Successful knowledge management projects, Sloan Management Review, 39 (2), pp. 43–57.
Dekker, S. W. A. (2005): Ten Questions About Human Error, A New View of Human Factors and System Safety, Human Factors in Transportation, Lawrence Erlbaum Associates, Mahwah, NJ.
Downey, G., Dalidowicz, M., and Mason, P. H. (2015): Apprenticeship as method, embodied learning in ethnographic practice, Qualitative Research, 15 (2), pp. 183–200.
EFPIA (2010): The Pharmaceutical Industry in Figures, European Federation of Pharmaceutical Industries and Associations, Brussels, available at: https://www.efpia.eu/media/15488/the-pharmaceutical-industry-in-figures-edition- 2010.pdf, accessed 18 April 2020.
Endsley, M. R., and Robertson, M. M. (1996): Team Situation Awareness in Aviation Maintenance, Proceedings of the Human Factors and Ergonomics Society Annual Meeting, 40 (21), pp. 1077–1081.
Fischer M. and Röben, P. (2001): Ways of Organizational Learning in the Chemical Industry and their Impact on Vocational Education and Training, A Literature Review, ITB, Bremen.
Fischer, M, Boreham, N. and Nyhan, B. (2004): European perspectives on learning at work, the acquisition of work process knowledge, Cedefop reference series no. 56, Office for Official Publications of the European Communities, Luxembourg.
Fischer, M. (2005): The integration of work process knowledge into human resources development, Human Factors and Ergonomics in Manufacturing, 15 (4), pp. 369–384.
Fischer, M. and Röben, P. (1997): Arbeitsprozeßwissen im chemischen Labor, Arbeit, Zeitschrift für Arbeitswissenschaft, Arbeitspsychologie und Arbeitsgestaltung, 3 (6), pp. 247–66.
Fischer, M. and Röben, P. (2002b): Cases of Organizational Learning in the Chemical Industry, An Empirical Study, ITB, Bremen.
Fischer, M., and Röben, P. (2002a): The work process knowledge of chemical laboratory assistants, in: Boreham, N., Fischer, M., and Samurcay, R. (ed.), Work Process Knowledge, London, Routledge, pp. 40–55.
Food and Drug Administration (2004): U.S. Pharmaceutical CGMPs for the 21st Century, A Risk-Based Approach, U.S., Maryland, available at: https://www.fda.gov/media/77391/ download, accessed 7 March 2020.
Friberg, T. (2017): The (Re)making of Flow, Mediator Companies and Knowledge Production, Journal of Business Anthropology, 6 (2), pp. 199-217.
Gad, S. (2008): Pharmaceutical Manufac- turing Handbook: Production and Processes, John Wiley & Sons, Hoboken, NJ.
Gavish, N., Gutiérrez, T., Webel, S., Rodríguez, J., Peveri, M., Bockholt, U., and Tecchia, F. (2015): Evaluating virtual reality and augmented reality training for industrial maintenance and assembly tasks, Interactive Learning Environments, 23 (6), pp. 778–798.
Gieryn, T. (2002): What buildings do, Theory and Society, 31 (1), pp. 35–74.
Goodman, E., Moed, A., and Kuniavsky, M. (2012): Observing the User Experience, A Practitioner’s Guide to User Research, Morgan Kaufmann, Burlington, MA.
Grudin, J. (2017): From Tool to Partner: The Evolution of Human-Computer Interaction, Synthesis Lectures on Human-Centered Informatics, Morgan and Claypool Publishers, Pennsylvania.
Grundgeiger, T., Hester, J., Held, V., and Hurtienne, J. (2017): Beyond knowledge acquisition, Medical device training as a cooperative process, in: Companion of the 2017 ACM Conference on Computer Supported Cooperative Work and Social Computing, 2017, ACM, New York, NY, pp. 187-190.
Haile, E. L., Taye, B., and Hussen, F. (2012): Ergonomic Workstations and Work-Related Musculoskeletal Disorders in the Clinical Laboratory, Laboratory Medicine, 43 (2), pp. 11-19.
Hajou, A., Batenburg, R., and Jansen, S. (2014): How the Pharmaceutical Industry and Agile Software Development Methods Conflict, A Systematic Literature Review, in: Proceedings – 14th International Conference on Computatio- nal Science and Its Applications ICCSA, 2014, pp. 40-48.
Harrington, J. (2013): A predicament, animal models and human tissue in medical research, Configurations, 21 (2), pp. 183–200.
Hartson, R., Pyla P. S. (2019): The UX Book, Agile UX Design for a Quality User Experience, Elsevier LTD, Oxford.
Hockey, G. R. (2005): Operator functional state, the prediction of breakdown in human performance, in: J. Duncan, L. Philips, and P. McLeod (ed.), Measuring the Mind: Speed, con- trol, and age, Oxford University Press, New York, pp. 373–395.
Holtzblatt, K., and Beyer, H. R. (2017): Contextual Design, Design for Life, Elsevier, Morgan Kaufman, Cambridge, MA.
Hunter, J. and Stephens, S. (2010): Is open innovation the way forward for big pharma? Nature Reviews Drug Discovery, 9, pp. 87-88.
Hutchinson, R. and M. Zain (2009): Internal audit quality, audit committee independence, growth opportunities and firm performance, Corporate Ownership and Control 7 (2), pp. 50- 63.
ISO/IEC stage 13407: Human-Centered Design Processes for Interactive Systems, 1999.
Jakl, A., Schöffer, L., Husinsky, M., and Wagner, M. (2018): Augmented reality for industry 4.0, Architecture and user experience, in: CEUR Workshop Proceedings, 2299, pp. 38–42.
Jones, PH. (2005): Information practices and cognitive artifacts in scientific research, Cogn. Technol. Work, 7 (2), pp. 88–100.
Jones, PH., Nemeth, CP. (2004): Cognitive artifacts in complex work, in: Ambient intelligence for scientific discovery Conference, foundations, theories, and systems, 2004, pp. 152– 183.
Jordan, B., Lambert, M. (2009): Working in Corporate Jungles: Reflections on Ethnographic Praxis in Industry, in: M. Cefkin (ed.), Ethnography and the corporate encounter, Reflections on Research in and of Corporations, Berghahn Books, pp. 95-133.
Kant, V., and Burns, C. M. (2016): Engaging nanotechnology, ethnography of lab-on-a-chip technology in small-scale fluidics research, Cognition, Technology and Work, 18 (1), pp. 33–52.
Kanza, S., Willoughby, C., Gibbins, N., Whitby, R., Frey, J. G., Erjavec, J., Kovač, K. (2017): Electronic lab notebooks, can they replace paper? Journal of Cheminformatics, 9 (1), pp. 1–15.
Karim, R., and Tretten, P. (2014): Enhancing the usability of maintenance data manage- ment systems, Journal of Quality in Maintenan- ce Engineering, 20 (3), pp. 290–303.
Knorr Cetina, K. (1995): Laboratory Studies, the Cultural Approach to the Study of Science, in: Jasanoff S., Markle G., Petersen J. and Pinch T. (ed.), Handbook of Science and Technology Studies. Thousand Oaks, CA, Sage, pp. 140–166.
Konstantinos, K., Themistocleous, M., Byrne, P., Ross, D., Cromie, S., and Corrigan, S. (2011): Investigating Human Factors in Biotechnology and Pharmaceutical Manufacturing industries, European, Mediterranean and Middle Eastern Conference on Information Systems, 2011, At Athens, pp. 294-305.
Kruse, W. (1986): On the necessity of labour process knowledge, in: J. Schweitzer (ed.), Training for a human future, Juventa, Weinheim, Basel, pp. 188–193.
Kruse, W. (1986): Von der Notwendigkeit des Arbeitsprozeß-Wissens, in: J. Schweitzer (ed.), Bildung für eine menschliche Zukunft, Juventa- Verlag, Weinheim, Basel, pp. 188–193.
Kujala, S., Kauppinen, M., and Rekola, S. (2001): Bridging the gap between user needs and user requirements, Advances in Human- Computer Interaction I, Proceedings of the Pan- hellenic Conference with International Participation in Human-Computer Interaction PC-HCI, 2001, pp. 45–50.
Kuselman, I., Pennecchi, F., Fajgelj, A., and Karpov, Y. (2013): Human errors and reliability of test results in analytical chemistry, Accreditation and Quality Assurance, 18 (1), pp. 3–9.
Ladner, Sam (2014): Practical Ethnography, A Guide to Doing Ethnography in the Private Sector, Left Coast Press, New York.
Latour, B. and Woolgar, S. (1986 [1979]): Laboratory Life, the Construction of Scientific Facts,
Lee, S. T., Kim, S. Y., and Gilmore, D. (2017): Human-in-the-loop evaluation of human-machine interface for power plant operators, in: IEEE International Conference on Systems, Man, and Cybernetics (SMC), 2017, pp. 34–39.
Lee, S.L., O’Connor, T.F., Yang, X. et al (2015): Modernizing Pharmaceutical Manufacturing, from Batch to Continuous Production, Journal of Pharmaceutical Innovation, 10, pp. 191-199.
Lewis J, Hughes J. and Atkinson, P. (2014): Relocation, realignment and standardization, circuits of translation in Huntington’s Disease, Social Theory and Health, 12 (4), pp. 396–415.
Lindgaard, G., Dillon, R., Trbovich, P., White, R., Fernandes, G., Lundahl, S., and Pinnamaneni, A. (2006): User needs analysis and requirements engineering, theory and practice, Interacting with Computers, 18 (1), pp. 47–70.
Lischke, L., Mayer, S., Preikschat, A., Schwei- zer, M., Vu, B., Wočniak, P. W., and Henze, N. (2018): Understanding Large Display Environments, Contextual Inquiry in a control room, Extended Abstracts of the CHI Conference on Human Factors in Computing Systems, 2018, pp. 1–6.
Lynch, M. (1985): Art and Artifact in Laboratory Science, A Study of Shop Work and Shoptalk in a Research Laboratory, Studies in Ethnomethodology, London, Routledge.
Maclean, R., Wilson, D., and Chinien, C. (2009): International Handbook of Education for the Changing World of Work, Bridging Academic and Vocational Learning. Springer, Science+Business Media B.V., Dordrecht.
Mariani, M. (2002): Work process knowledge in a chemical company, in: Boreham, N., Fischer, M., and Samurcay, R. (ed.), Work Process Knowledge, London, Routledge, pp. 15–25.
Mayring, P. (2000): Qualitative Content Analysis, Qualitative Social Research, 1 (2).
Mohler, W. (1970): Der Laborant. Entstehung und Entwicklung eines Berufes in der Baseler chemischen Industrie. Verlag Herbert Lang, Bern.
Myerson, A.S., Krumme, M., Nasr, M., Thomas, H., Braatz, R.D (2015): Control systems engineering in continuous pharmaceutical manufacturing, Journal of Pharmceutical Sciences, 104 (3), pp. 832-839.
Nielsen, J. (1995): Usability Engineering, Academic Press, San Diego, CA.
Norman, D. (1988): The Design of Everyday Things, Basic Books, New York.
O’Dell, C. and Grayson, C.J. (1998): If only we knew what we know, Identification and transfer of internal best practices, California Management Review, 40 (3), pp. 254–74.
Osakwe, O. (2016): Pharmaceutical Formula- tion and Manufacturing Development: Strategies and Issues, in: Osakwe, O. and Rizvi, S. (ed.), Social Aspects of Drug Discovery, Development and Commercialization, pp.169-187
Palviainen, J., and Leskinen, H. (2006): User Research Challenges in Harsh Environments, A Case Study in Rock Crushing Industry, in: A. G. Nilsson, R. Gustas, W. Wojtkowski, W. G. Wojtkowski, S. Wrycza, and J. Zupančič (ed.), Advances in Information Systems Development, Springer, Boston.
Penders, B., Verbakel, J. M. A., & Nelis, A. (2009): The Social Study of Corporate Science, A Research Manifesto, Bulletin of Science, Technology & Society, 29 (6), pp. 439–446.
Perrow, C. (1983): The Organizational Context of Human Factors Engineering, Administrative Science Quarterly, 27, pp. 521-541.
Perry, J., and Baum, J. (2018): Assessing the Laboratory Environment, in: ACS Symposium Series, Accessibility in the Laboratory, (1272), pp. 3–25.
Plowman, T. (2003): Ethnography and Critical Design Perspective, in: B. Laurel (ed.), Design Research, MIT Press, London, pp. 30–38.
Preece, J., Rogers, Y., and Preece, J. (2007): Interaction design, Beyond human-computer interaction, Wiley, Chichester.
Privitera, M.B. (2015): Contextual Inquiry for Medical Device Design. Elsevier, Oxford.
Privitera, M.B. (Ed.). (2019): Applied Human Factors in Medical Device Design, Elsevier, London.
Reason, J. (1990): Human Error, Cambridge University Press, New York.
Rogers, E. M. (2003): Diffusion of Innovations, Free Press, New York.
Ross, P. E. (2008): Ergonomics Improving Safety in the Laboratory Environment, Professi- onal Safety, 53 (8), pp. 6.
Schmauderer, E. (1973): Der Chemiker im Wandel der Zeit. Skizzen zur geschichtlichen Entwicklung des Berufsbildes, VCH, Weinheim.
Schmitt, L. (2019): Challenges affecting the adoption of B2B electronic marketplaces, Jour- nal of Business Chemistry, 2019 (3), pp. 154-164.
Schneiderman, B. (2016): The New ABCs of Research, Achieving Breakthrough Collaborati- ons, Oxford University Press, Oxford.
Schuhmacher, A., Germann, P., Trill, H., Gass- mann, O. (2013): Modes for open innovation in the pharmaceutical industry, Drug Discovery Today, 18, pp. 23-24.
Seligman, Martin E. P.; Csikszentmihalyi, Mihaly (2000): Positive Psychology, An Intro- duction, American Psychologist, 55 (1), pp. 5–14.
Sharit, J. (2006): Human Error, in: Salvendy, G. (ed.), Handbook of Human Factors and Ergo- nomics, John Wiley and Sons, Hoboken, New Jersey, pp. 708-760.
Singh, A. (2019): Understanding User Adaptation to New Technology in the Manufacturing Industry, User Experience Magazine, 19 (1).
Sørensen, K. B., Christiansson, P., Svidt, K., Jacobsen, K., and Simoni, T. (2008): Radio Frequency Identification in Construction Operation and Maintenance, Contextual Analysis of User Needs, in: A. Ren, Z. Ma, and X. Lu (ed.), Proceedings of 12th International Conference on Computing in Civil and Building Engineering and 2008 International Conference on Information Technology in Construction, 2008.
Sormani, P. (2014): Respecifying Lab Ethnography, London and New York, Routledge.
Stanton, N., Salmon, P., Rafferty, L., Walker, G., Baber, C., and Jenkins, D. (2013): Human Fac- tors Methods, CRC Press, London.
Stephens, N., and Lewis, J. (2017): Doing la- boratory ethnography, reflections on method in scientific workplaces, Qualitative Research: QR, 17 (2), pp. 202–216.
Stephens, N., Atkinson, P., and Glasner, P. (2008): The UK Stem Cell Bank as Performative Architecture, New Genetics and Society, 27 (2), pp. 87–99.
Storz, P., Fries, M., and Klöden, W. (1997): Change of Work in The Scientific Laboratory, in: K. Drechsel, P. Storz, and G. Wiesner (ed.), Work – Education – Vocation (13th ed., pp. 148). Dres- den, Germany: wbw Verlag and Mediaservice für Wissenschaft, Bildung, Wirtschaft.
Storz, P., Fries, M., and Klöden, W. (1997): Change of Work in The Scientific Laboratory, in: K. Drechsel, P. Storz, and G. Wiesner (ed.), Work – Education – Vocation, wbw Verlag and Media- service für Wissenschaft, Bildung, Wirtschaft, Dresden.
Stuart K. Card, Allen Newell, and Thomas P. Moran (1983): The Psychology of Human-Computer Interaction. L. Erlbaum Assoc. Inc., Hillsdale, NJ, USA.
Talanquer, V. (2006): Commonsense Che- mistry, A Model for Understanding Students’ Alternative Conceptions, Journal of Chemical Education, 83 (5), pp. 811.
Terzic, I., Zoitl, A., Rooker, M., Strasser, T., Vrba, P., and Mačík, V. (2009): Usability of Multiagent Based Control Systems in Industrial Automation, in: V. Mačík, T. Strasser, and A. Zoitl, (ed.), Holonic and Multi-Agent Systems for Manufacturing, Springer, Berlin Heidelberg.
Thimbleby, H. (2019): Three laws for paperlessness, Digital Health, 5, available at: https:// journals.sagepub.com/doi/full/10.1177/2055207619827722, accessed 28 February 2020.
Thrift, N. (2006): Reinventing invention, new tendencies in capitalist commodification, Economy and Society, 35 (2), pp. 279–306.
Throckmorton, D. (2014): Examining Drug Shortages and Recent Effort to Address Them, Statement to the House of Representatives, Subcommittee on Health, Committee on Energy & Commerce, available at: https:// www.govinfo.gov/content/pkg/CHRG-113hhrg88610/pdf/CHRG-113hhrg88610.pdf, accessed 7 March 2020.
Torz, P., and Eichhorn, S. (2001): Organizational Learning and Shaping in SMEs from the Chemical Industry, in: K. Drechsel, P. Storz, and G. Wiesner (ed.), Work – Education – Vocation, wbw Verlag and Mediaservice für Wissen- schaft, Bildung, Wirtschaft, Dresden.
Traweek, S. (1988): Beamtimes and Lifetimes, The World of High Energy Physicists, Harvard University Press, Cambridge.
Ulbrich, M., Aggarwal, V. (2019): The Digital Revolution is coming to chemical laboratories, Journal of Business Chemistry, 2019 (2), pp. 76- 81.
Wang, W. Y. C., Heng, M. S. H., and Chau, P. Y. K. (2010): The adoption behavior of information technology industry in increasing business- to-business integration sophistication, Information Systems Journal, 20 (1), pp. 5–24.
Werner, S., and Kirsten, B. (2003): Ethnographic Fieldwork Under Industrial Constraints, Toward Design-in-Context, International Journal of Human-Computer Interaction, 15 (1), pp. 41–50.
Williams, S. J., Gabe, J., and Davis, P. (2008): The sociology of pharmaceuticals, Progress and prospects, Sociology of Health and Illness, 30 (6), pp. 813–824.
World Bank Group (1998): Pollution Preven- tion and Abatement Handbook, Pharmaceuticals Manufacturing. Project Guidelines, Industry Sector Guidelines.
Yi, Zhixian. (2018): Techniques for Identifying Users’ Needs and Wants, in: Zhixian, Yi (ed.), Marketing Services and Resources in Information Organizations, Chandos Publishing, Cambridge, MA, pp. 29-37.
Appendix
Complete list of elicited user needs:
Scientist:
(1) A scientist in this laboratory needs to proof his thinking around a concrete set of chemistry. Therefore, he needs to produce an understanding of the process, that is, about the reaction time and the mixing characteristics of the equipment. With these data a scientist in this laboratory needs to build a model to be able to scale that in correlation to bigger setups for mass production.
Continuous Processing Technician (Technician):
Design Phase
(2) A technician in this laboratory needs a dia- gram of the setup in order to understand what a scientist wants to do.
(3) A technician in this laboratory needs to in- terpret process design drawings in order to be able to start building the setup
(4) A technician in this laboratory needs to identify changed parts of the setup diagram (in his proforma) in order to account for the ne- west version of the setup.
(5) A technician in this laboratory needs to read and understand the process design/drawing in order to know which parts he has to find for the setup.
(6) A technician in this laboratory needs to know the “Inlets” and “Outlets” of a setup in order to know how special components are connected to the other parts of the setup.
(7) A technician in this laboratory needs to get as much information about the setup as pos- sible from different people in order to understand their goals.
(8) A technician in this laboratory needs to get specific information on layers and details of setup diagram in order to avoid iterations.
(9) A Technician in this laboratory needs to be able to understand the necessities of process design iterations in order to avoid knowledge gaps and frustration.
(10) A Technician in this laboratory needs more detailed and standardized process diagram containing consistent symbols in order to be more efficient.
Setup Phase
(11) A technician in this laboratory needs to visu- ally control the setup flow in order to get a first impression of its status.
(12) A technician in this laboratory needs to be aware and constantly control setup equipment for errors, especially pumps, in order to be able to change and thereby dispose the defective equipment.
(13) A technician in this laboratory needs to flush pumps with solvents in order to clean setup parts.
(14) A technician in this laboratory needs to be able to put a component aside for further cleaning when it is not clean and should be able to take a new (clean) component instead in order to continue a setup.
(15) A technician in this laboratory needs to identify errors on the operation system gathering data on the setup.
(16) A technician in this laboratory needs to be able to build setups in iteration in order to coordinate/compensate missing equipment.
(17) A Technician in this laboratory needs to do (besides visual check, a pressure check) a flow rate check with equipment in order to assess if the pumps are delivering what their status claims to be able to deliver.
(18) A Technician in this laboratory needs to be able to work on basis of the process diagram without thinking of alternative setup possibilities including special components.
(19) A Technician in this laboratory needs to be able to work with a sorted and fully equipped (all in one) toolbox in order to be able work “out of the box” spontaneously.
(20) A Technician in this laboratory needs to be reassured about working rules for each component, with or without serial number, in order to know how to document/log them discuss original setup designs/drawings/plans at the very beginning in order to get a clear understanding of the goals.
(21) A Technician in this laboratory needs to be able to help himself in terms of training, that is, getting specific manufacturer manuals on the spot.
(22) A Technician in this laboratory needs to analyse the setup step by step with his boss or a chemist before he is allowed to run the setup.
(23) A Technician in this laboratory needs to have an equipment-recipe attached to the process design/drawing in order be more efficient.
(24) A Technician in this laboratory needs to be aware of component capabilities and weaknesses that can unfold during instalment of different components (e.g. the pressure or tension of a screw that is fixing a tube that might lead to a leak).
Communication
(25) A technician in this laboratory needs to communicate to colleagues in order to seek and get advice or to find the right equipment.
(26) A technician in this laboratory needs to be able to talk to a chemist engineer in order to start the cleaning of components.
(27) A technician in this laboratory needs to or- der a specialist (e.g. from IT or facility management) to restart and fix the operation system in order to keep the process running.
(28) A technician in this laboratory needs to react spontaneously to demands of scientists who are present in the lab and adapt his plans or priorities.
(29) A technician in this laboratory needs to discuss original setup designs/drawings/plans at the very beginning in order to get a clear understanding of the goals.
(30) A technician in this laboratory needs to be able to commit suggestions for setup changes or adaptations in order to start a discussion with a scientist (e.g. about how the setup might be more efficient).
(31) A technician in this laboratory needs to be aware of the intranet team-sites in order look up suggestions from colleagues and share his own experiences with specific equipment or setups.
(32) A Technician in this laboratory needs to communicate with his colleagues to share knowledge about current status of a setup and what is to do next.
(33) A Technician in this laboratory needs to know the current status of project setups, who is working right now, who will be working in the next shift (am-shift & pm-shift) and when who will be on vacation.
(34) A Technician in this laboratory needs to anticipate support by colleagues in order to know how to wrap up his shift or how to leave a setup (turn off or leave it running).
(35) A Technician in this laboratory needs to be able to place orders for equipment sooner in order to avoid the lack of equipment.
(36) A Technician in this laboratory needs to be able to share his current point of view of a part of the setup in order to get ‘hands-on’ feedback or advice from a colleague.
(37) A Technician in this laboratory needs to be reassured about his role in order to manage expectations regarding rotation of team mem- bers and responsibilities.
Documentation
(38) A technician in this laboratory needs to get a proforma to be able to focus on the required setup specifications.
(39) A technician in this laboratory needs to know where specific parts of equipment are right now in order to collect them to be able to start the setup process.
(40) A technician in this laboratory needs to document every adaptation of the setup dia- gram in the Proforma.
(41) A technician in this laboratory needs to be aware that there might be a lot of changes to the setup and the Proforma in order to optimize the process that is to be developed.
(42) A technician in this laboratory needs to understand protocols for lab notebooks and discuss them with an engineer.
(43) A technician in this laboratory needs to visually inspect components and take them apart while documenting their status for cleaning.
(44) A technician in this laboratory needs to fill out a cleaning record that is a protocol within the lab notebook that requires him to sign of single tasks, in order to be accountable for the cleaning of components.
(45) A technician in this laboratory needs to know in which condition a component is before he is allowed to use it.
(46) A technician in this laboratory needs to sign things correctly in the lab notebook, that is, starting on the actual protocol and ending on the page of the lab notebook that contains the actual protocol.
(47) A technician in this laboratory needs to be aware of several projects, their status and current tasks.
(48) A technician in this laboratory needs to gather and document all information regarding his current tasks in order to hand it over to colleagues during the handover of a shift.
Operations Team Leader:
(49) An Operations Team Leader in this laboratory needs to organize and manage the storage and maintenance of materials and equipment in order to keep the workflow of the lab running.
(50) An Operations Team Leader in this labora- tory needs to prioritize many different requests.
(51) An Operations Team Leader in this laboratory needs to maintain the lab’s intranet team- site in order to track tasks and equipment, e.g. in combination with Proformas.
(52) An Operations Team Leader in this laboratory needs to document equipment status, that is, where they are in use at the moment, their current status as material and where it is stored.
(53) An Operations Team Leader in this laboratory needs to combine information from lab notebooks and Proformas in order to keep track of equipment.
(54) An Operations Team Leader in this laboratory needs to manage single requests, that is, assigning equipment to the request by checking its status (e.g. via tracking-list or spread-sheet).
(55) An Operations Team Leader in this laboratory needs to think about ways to track equipment with barcodes.
(56) An Operations Team Leader in this laboratory needs to talk to scientists in order to discuss the organization of resources for new ideas and significant setup changes.
(57) An Operations Team Leader in this laboratory needs to prepare Proformas and assign them to technicians.
(58) An Operations Team Leader in this laboratory needs to relate changes of shifts, the over- view and current status information for each project and the current status of individual Proformas.
(59) An Operations Team Leader in this laboratory needs to know if the setup is GMP or Not- GMP and identify relevant implications.
(60) An Operations Team Leader in this laboratory needs to be able to standardize resource management in order to plan and structure the expectations of team members.
(61) An Operations Team Leader in this laboratory needs to be able to set requests ‘on hold/ halt’ in order to avoid excessive demands by gathering more information and discussing challenges.
(62) An Operations Team Leader in this laboratory needs to organize requests in coordination between email and demands that are broadcasted between team members and in the lab, that is, e.g. on a whiteboard.
(63) An Operations Team Leader in this laboratory needs to coordinate the skills of technicians to tasks and further discuss demands for training.
(64) An Operations Team Leader in this laboratory needs to be able to broadcast specific information for safety issues, e.g. prior experiences or highlight hazards working with a specific component.
(65) An Operations Team Leader in this labora- tory needs to broadcast the following key information for setups: What equipment, what capabilities for the materials of construction (e.g. stainless steel), where is it going, what are the goals to aim for with this setup.
(66) An Operations Team Leader in this laboratory needs to be able to manage/coordinate initial meetings including technicians in order to enable an ongoing knowledge transfer between science and know-how.
(67) An Operations Team Leader in this laboratory needs to be able to highlight and coordinate differences between the workstyles of GMP and NON-GMP.
Continuous Processing Engineer (Engineer):
(68) An engineer in this laboratory needs to be able to proof the feasibility of experiments.
(69) An engineer in this laboratory needs to be able to work and easily switch between the office work place and the laboratory in order to discuss process theory and feasibility.
(70) An engineer in this laboratory needs to be aware of operating conditions, flow rate and volume of materials on the one side and suitab- le equipment on the other side.
(71) An engineer in this laboratory needs to wri- te specification sheets for materials.
(72) An engineer in this laboratory needs to be able to check the suitability of equipment by reading manufacturer manuals and so-called ‘specification sheets’ which contains basic information of materials and limits.
(73) An engineer in this laboratory needs to check the Team-Sites/Spreadsheets to see which equipment is available.
(74) An engineer in this laboratory needs shared templates for process drawings.
(75) An engineer in this laboratory needs to plan and draw process schematics on his office-PC and share these schematics with technicians at the place of the setup.
(76) An engineer in this laboratory needs to be able to use small-scale experiment setups for simulation in order to brainstorm and discuss possible setup solutions.
(77) An engineer in this laboratory needs to know which special components are ready for setup and if not, what they need to become ready for use (e.g. pressure sensors and tempe- rature sensors require an interface with a con- trol and data logging system.
(78) An engineer in this laboratory needs to know and document the status of each compo- nent before using it, that is, the history of it including dates of use, project/experiment number, name of the person who has signet out, specific logging codes for the component.
(79) An engineer in this laboratory needs to document smaller changes and adaptations in the lab notebook (“logging”) using references via logging codes (shortcuts) and specific serial numbers for specific components like pumps that are enlisted in the lab notebook.
(80) An engineer in this laboratory needs to document events in detail during experiments or cleaning.
(81) An engineer in this laboratory needs to be able to simplify documentation habits, especi- ally in order to document group meetings.
(82) An engineer in this laboratory needs to know risks/hazards about chemicals as well as compatibility data of the equipment (high temperatures & pressures) in order to get an idea about the process assessment.
(83) An engineer in this laboratory needs to get a rough idea of what has to happen/to be done in what order to document processes.
(84) An engineer in this laboratory needs to be able to place short notes/instructions on the setup in order to highlight simple tasks, e.g. cleaning of a component.
(85) An engineer in this laboratory needs to be able to communicate instructions for the evening shifts in order to prepare things for the next morning.
(86) An engineer in this laboratory needs to know the weak spots of a setup, that is, what is the limiting bit of equipment and be able to focus on the ‘delivery system’ and the ‘reactor’.
(87) An engineer in this laboratory needs to identify a component by serial number in order to discriminate between logged equipment and consumable materials.
(88) An engineer in this laboratory needs to know about the current stock spares and who is involved in the ordering of new equipment.
(89) An engineer in this laboratory needs to be sure about the documentation and specific storage of components.
(90) An engineer in this laboratory needs to know the last person responsible for a component of equipment.
(91) An engineer in this laboratory needs to know compatibility of solvents, max. and min. pressure and temperature rating for equipment in order to build a setup for experimentation.
(92) An engineer in this laboratory needs to know the last settings of components, e.g. of pressure reliefs, and how to change the settings the right way (and when it was tested).
(93) An engineer in this laboratory needs to decide on which reactor to use, inlet-points and their class as well as the outlet-points in order to start modelling a small-scale simulation setup with the based on a rough idea for a process.
(94) An engineer in this laboratory needs to combine the information of operation conditions (temperatures), what and where to operate, problems with operation limits, operation capacities of the system (heat & cold), needed speed of mixer-components.
(95) An engineer in this laboratory needs to know the maximum pressure of the pump compared to the system pressure, also considering flow rate.
(96) An engineer in this laboratory needs to be able to find the person to talk to in order to gather information about new components.
Interview guideline for contextual inquiry in continuous manufacturing lab
Introduction / Instruction
The goal of this document is to guide you through the data gathering process during user interviews on site. Overall, we aim to cover and understand the sense making processes of people during work within a specific work place that comes with a specific culture, including respective individual and social practices in place (e.g. habits, routines, and rules).
The guideline document structure follows a successive logic, beginning with easy to answer questions for rapport to warm-up or break the ice for having a relaxed and authentic discussion. Interviewees shall know and understand that this is not a test, that we are just curious outsiders trying to understand the way they think and work.
The overall structure is as follows:
1. Introduction / Instruction
2. Participant & his goals (“Me as a person & employee”)
3. Working in a team („Me and the others in the team”)
4. Tasks („What I do & my tools“)
5. General workflow („What I usually do, in which order and why”)
6. Current workflow (Introduction to work place)
7. Closing the interview